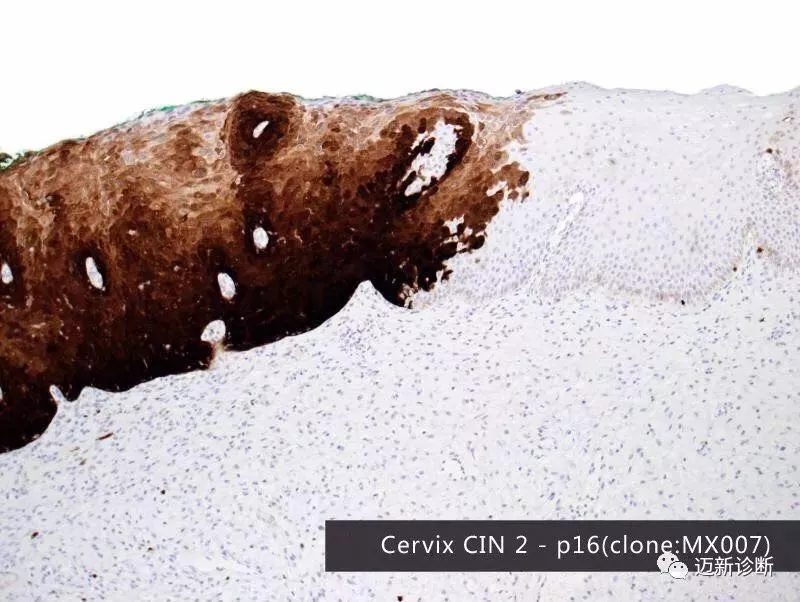
Third-party Independent Laboratory Collaborates with Maixin on p16/Ki-67 Dual Staining Detection to Comprehensively Tap into the Billion-Dollar Cervical Cancer Screening Market
Recently, numerous third-party independent laboratories have successively reached strategic cooperation intentions with Maixin Reagents, which registered and approved the p16/ki-67 detection kit on March 1, jointly promoting the “Cervical Cancer Screening Upgrade Package Project” (TCT+HPV+p16/Ki-67 dual staining detection) at third-party independent laboratories’ testing centers, branches, cooperative joint laboratories, and their affiliated companies nationwide, comprehensively deploying in the billion-level cervical cancer screening market.

Since 2009, China has included the “Two Cancers” (cervical cancer and breast cancer) screening project for rural women as a major public health project in healthcare reform. By 2017, a cumulative total of 70 million rural women had undergone cervical cancer screening. Over the eight years, 97,062 cases of cervical invasive cancer and precancerous lesions were detected, achieving significant social benefits and economic effects.
Third-party independent laboratories are a vital force in the national cervical cancer screening project and an important component in urban cervical cancer screening. The cervical cancer screening projects, such as TCT or TCT+HPV detection, have become a significant part of the pathological testing services offered by third-party independent laboratories.
The third-party independent laboratories that have reached agreements with Maixin have added p16/ki-67 dual staining detection to their existing cervical cancer screening packages, launching an upgraded package consisting of TCT+HPV+p16/Ki-67dual staining, forming a comprehensive solution for cervical cancer screening. For potential high-grade lesions, dual staining has sensitivity comparable to HPV testing but with specificity doubled, significantly reducing false-positive results and effectively decreasing the colposcopy referral rate by 50%. Additionally, dual staining shows high sensitivity of 89.3% and 94.1% for diagnosing CIN2+ and CIN3+, respectively. In women with negative cytology but positive HPV results, Maixin’s p16/Ki-67 dual staining demonstrates high sensitivity and specificity, with sensitivity for CIN2+ reaching 90.9% and for CIN3+ reaching 100%.
The new package solution involves one sample collection, two specimens, and three reports, effectively addressing various methodological shortcomings, poor inter-observer reproducibility, insufficient quality control, and lack of technical personnel in current cervical cancer screening processes. The p16/Ki-67 dual staining detection, due to its simple interpretation and clear triage significance, brings cervical screening closer to the gold standard, playing a positive role in preventing cervical cancer, early diagnosis, early treatment, and pre-vaccination testing.
The p16 gene is a typical representative of cyclin-dependent kinase inhibitors. Overexpression of p16 indicates that cells are in the cell cycle arrest phase and can serve as a marker for cervical lesions. Ki-67 is a nuclear antigen gene marking cell cycle progression and proliferating cells, with its overexpression indicating cells are in the proliferation phase. Simultaneous overexpression of p16 and Ki-67 suggests dysregulation of the cell cycle. Detecting both p16 and Ki-67 in the same cell can serve as a marker for cell cycle dysregulation, associated with carcinogenic transformation induced by high-risk HPV viruses, helping to identify true lesion cells independently of morphological examination results. A positive p16/Ki-67 test strongly suggests high-grade lesions, providing an objective detection indicator to distinguish women with potential high-grade lesions.
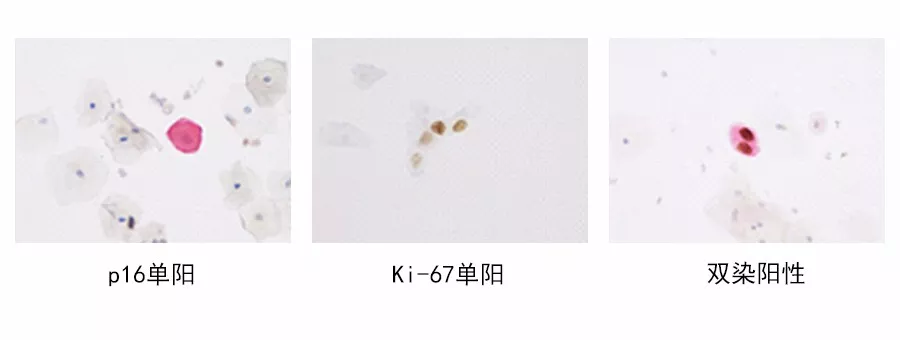
Compared to similar products, Maixin’s p16/Ki-67 detection kit offers lower manual operation reagent costs, faster processing, simpler interpretation, quicker slide reading, and higher positive detection rates, especially for borderline cells that are difficult to distinguish, making interpretation easier.
In 2014, building on Maixin’s successful development of p16, the company established the Immunocytochemistry Division, officially beginning market preparation for the dual staining kit. Over four years, comparisons and optimizations were conducted on various cytology preparation methods (Thinprep membrane method, BD sedimentation method, domestic natural sedimentation method, centrifugal smear method, etc.), and multicenter studies were carried out in Beijing, Fujian, Tianjin, Jiangxi, and Xinjiang to compare p16/Ki-67 dual staining with cytology, HPV testing, and histological results in parallel.
In 2015, Maixin’s p16/Ki-67 dual staining participated in the national “Appropriate Cervical Cancer Screening Technology and Demonstration Project for Rural China,” conducting a double-blind study of 3,000 people in Sichuan and Chongqing. From 2016 to 2017, it was invited to participate in academic exchanges at the World HPV Congress in Hong Kong and Cape Town, South Africa. All research data results were published in journals such as Cancer Prevention, Chinese Journal of Epidemiology, Chinese Journal of Oncology, and Chinese Journal of Pathology.
Professor Qiao Youlin from the Cancer Institute of the Chinese Academy of Medical Sciences, Professor Chen Guozhang from Hong Kong’s Queen Elizabeth Hospital, and Professor Yang Bin from the Cleveland Clinic in the United States have provided strong support and high praise for Maixin’s p16 and p16/Ki-67 dual staining: Maixin’s p16 and p16/Ki-67 dual staining kits are ideal auxiliary examination technologies for cervical cancer. They not only enable risk stratification and quantitative management of patients, balance screening sensitivity and specificity, and timely detect high-risk populations, reducing the harm from excessive colposcopy referrals, but are also simple and easy to implement with good health technology economics, representing a technological revolution in cervical cancer screening.