Striving for Excellence and Approaching Perfection, Maixin Achieves Outstanding Results Again in the NordiQC 2019 Interlaboratory Quality Assessment.
Editor’s Note:
NordiQC, the Nordic Immunohistochemistry Quality Control Center, is one of the internationally authoritative inter-laboratory quality assessment organizations for pathology. It has long been committed to promoting the standardization of immunohistochemistry processes and the optimization of experimental conditions. Maxin has consistently paid attention to its activities and actively participated in its related inter-laboratory quality assessment activities in recent years, achieving excellent results. We are pleased to announce that our company has once again achieved outstanding performance in this year’s NordiQC inter-laboratory assessment activities: General-Run55, Breast-Run B27, and HER2-Run H15. The evaluation results for HER2 (IHC/ISH), ER, SOX10, WT-1, CK5, Bcl-6, and ASMA antibodies were all excellent, successfully passing the assessment.
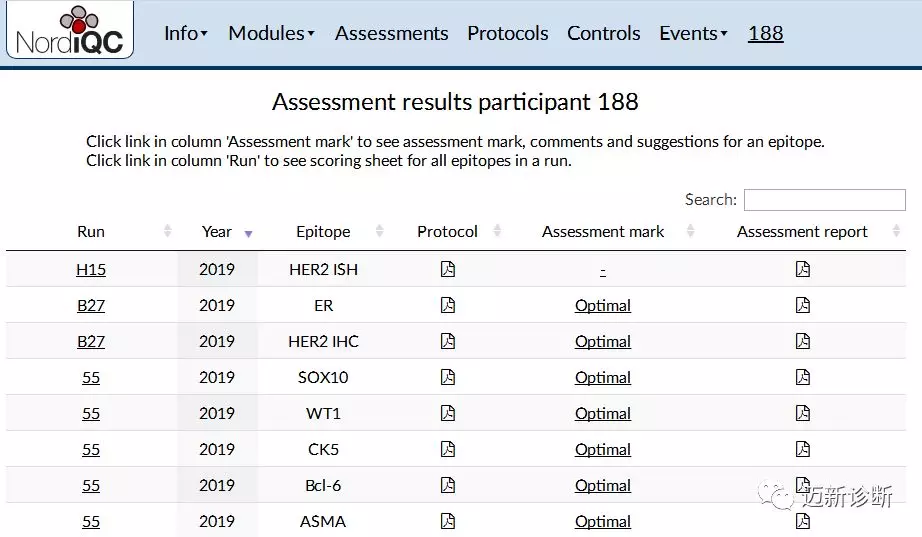
Figure 1. Maxin’s performance in the 2019 B27 and 55 rounds of immunohistochemistry inter-laboratory assessment activities

Figure 2. Maxin’s evaluation results in the 2019 H15 round of HER2 ISH assessment activity
It is worth mentioning that the WT1, HER2, and Bcl-6 antibodies are self-developed Maxin clone products. Since participating in NordiQC quality control in 2015,allthey have achieved excellent results,their outstanding performance and stable quality have withstood continuous testing, gaining recognition from international expert review organizations.
Subsequently, we will compile the performance of the above-mentioned Maxin antibodies participating inNordiQCinter-laboratory quality assessment activities (including staining images and antibody protocol recommendations) for your reference and selection. Please stay tuned!
