MUC4 is encoded by a gene located in the chromosomal region 3q21 (as shown in Figure 1). It is a transmembrane glycoprotein containing unique epidermal growth factor-like domains, exhibiting characteristics of both membrane-bound and secreted proteins (as shown in Figure 2): the expressed glycoprotein is located on the apical surface of mucosal epithelial cell membranes. Under normal conditions, it promotes epithelial cell renewal and differentiation, providing lubrication and protection for epithelial tissues. During carcinogenesis, the overexpression of MUC4 is associated with tumor metastasis and anti-apoptosis. It can be used in IHC diagnostic studies for various tumors such as pancreatic cancer, lung cancer, and endometrial cancer.
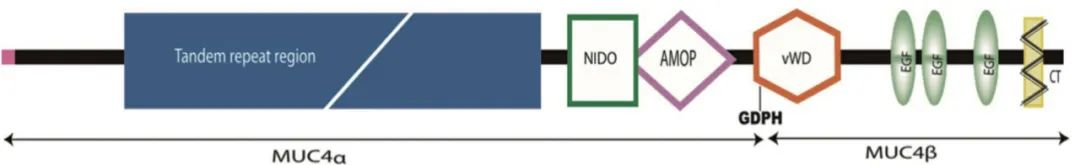
Figure 1. Schematic diagram of MUC4 structure (MUC4 can be cleaved at the GDPH site into MUC4α and MUC4β. MUC4α contains a variable number of tandem repeat regions, a NIDO domain, and an adhesion-associated (AMOP) domain. MUC4β contains one von Willebrand factor (vWD) domain, three epidermal growth factor (EGF)-like domains, one transmembrane domain, and a 22-amino acid cytoplasmic tail (CT). The NIDO, AMOP, and vWD domains are unique to MUC4 and are not found in other mucins.)
Figure 2. Expression and distribution of MUC4 (Note: Cited from the Genecards database (https://www.genecards.org/))
Role of Expression in Pancreatic Cancer
MUC4 expression is not present in normal pancreas or inflammatory diseases like chronic pancreatitis, but it can be detected in pancreatic cancer precursor lesions, and its expression level gradually increases with the progression of pancreatic cancer.
Jhala N et al. compared resected pancreatic cancer tissues (n=40) with fine-needle aspiration specimens (n=65) and found that MUC4 could adequately distinguish adenocarcinoma from reactive ductal epithelial cells in fine-needle aspiration specimens. In ductal adenocarcinoma, over 90% of tissue samples were reported as MUC4 positive (p<.001). The study by Zhu Peng et al. found that the positive expression rates of MUC4 in normal pancreatic tissue, pancreatic intraepithelial neoplasia (PanIN-1, PanIN-2, PanIN-3), and ductal adenocarcinoma were 0%, 17.4%, 52.6%, 84.6%, and 90.0%, respectively. Ansari et al. found a high concordance (82%) of MUC4 expression between primary and paired metastatic ductal adenocarcinoma lymph node tumors, suggesting that MUC4 expression is retained during metastasis and serves as a biomarker for this malignancy, useful for the early diagnosis of pancreatic precursor lesions and ductal adenocarcinoma.
Zhong Guodong et al. studied 168 cases of pancreatic cancer and found that the overall survival time of patients with high MUC4 expression was significantly lower than that of patients with low MUC4 expression. Kaplan-Meier analysis showed poor prognosis in patients with high MUC4 expression, and multivariate analysis indicated that high MUC4 expression was an independent factor for poor prognosis in pancreatic cancer patients, which is consistent with the findings of Zhu Peng et al.
Furthermore, MUC4 has been shown to mediate resistance to gemcitabine treatment. Research from the Fred & Pamela Buffett Cancer Center indicates that eliminating MUC4 expression reduces the proliferation and metastasis of pancreatic cancer cells and increases sensitivity to therapeutic agents, confirming the utility of MUC4 as a therapeutic target.
Role of Expression in Non-Small Cell Lung Cancer
Kwon et al. analyzed 343 cases of non-small cell lung cancer tissues and found that MUC4 expression rates were 81% in adenocarcinoma, 78% in squamous cell carcinoma, 75% in adenosquamous carcinoma, and 55% in large cell carcinoma, indicating the presence of MUC4 expression in non-small cell lung cancer, with positivity rates varying according to tumor histological type.
The study by Vishwa Jeet Amatya et al. found that MUC4 was expressed in spindle tumor cells of pulmonary sarcomatoid carcinoma (21/29, 72%) but not in any sarcomatoid mesothelioma (0/31, 0%). The sensitivity for distinguishing sarcomatoid mesothelioma from pulmonary sarcomatoid carcinoma was 100%, specificity was 72%, and accuracy was 87%, which is higher than markers like calretinin, D2-40, and Claudin-4. This is consistent with studies by Llinares, Mawas et al., suggesting that MUC4 can serve as a new marker for differentiating sarcomatoid mesothelioma from pulmonary sarcomatoid carcinoma.
Although the prognostic value of MUC4 in non-small cell lung cancer remains controversial, MUC4 has been utilized in the laboratory as a useful target antigen for targeted and immunotherapy in non-small cell lung cancer. Anti-MUC4 monoclonal antibodies possess properties that inhibit cancer cell growth and enhance the immune response of cancer cells to chemotherapy, suggesting that MUC4 IHC detection could become an important indicator for targeted therapy and immunotherapy in non-small cell lung cancer.
Role of Expression in Endometrial Cancer
Shi Xiaozhe et al. studied 76 cases of endometrial cancer tissues and 45 cases of normal endometrial tissues and found that the expression rates of MUC4 in endometrial cancer and normal endometrial tissues were 71.1% and 20.0%, respectively (p<0.01). Kaplan-Meier survival analysis showed that the survival time of patients with positive MUC4 expression was (38.48 ± 3.24) months, while that of patients with negative MUC4 expression was (55.57 ± 2.13) months (as shown in Figure 3), indicating that positive MUC4 expression suggests poor prognosis in endometrial cancer.
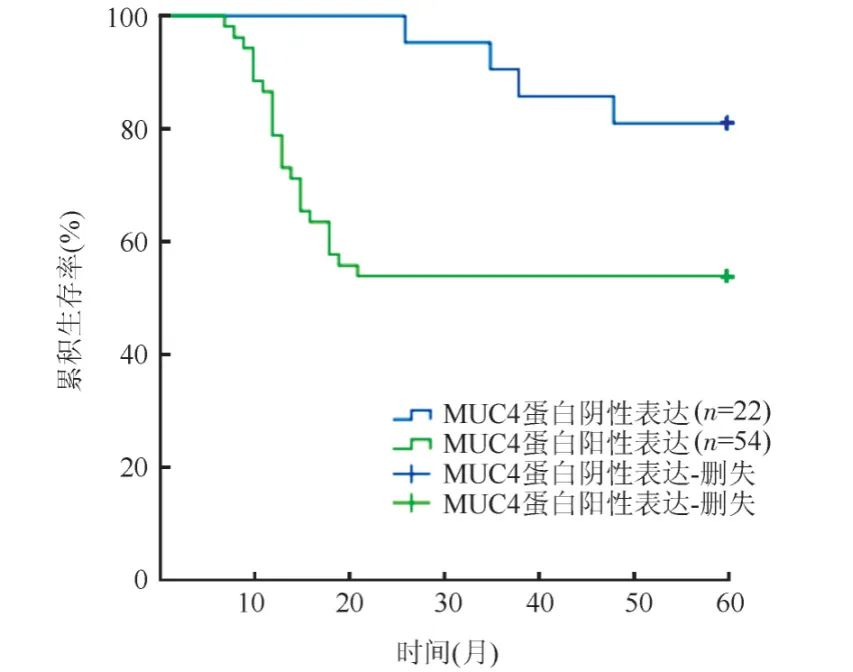
Figure 3. Kaplan-Meier analysis of the impact of MUC4 expression in endometrial cancer tissues on survival time
The IHC study by Zhao Ying et al. on 82 cases of endometrial cancer supports the above view. This study showed that the positive expression rate of MUC4 in endometrial cancer was significantly higher than in adjacent tissues. The progression-free survival rate of MUC4-positive patients was 24.56%, significantly lower than the 76.19% in negative patients. Cox proportional hazards regression model analysis indicated that MUC4 positivity is an important factor for poor prognosis in endometrial cancer.
Role of Expression in Other Tumors
The latest study by Nermeen M et al. indicates that MUC4 is a new diagnostic marker that can distinguish suspected meningioma from non-meningothelial tumors. The study by Benjamin Kaumeyer et al. also found that MUC4 expression is highly specific (90.9%) for BCR-ABL1+ and BCR-ABL1-like B-lymphoblastic leukemia, with high sensitivity for cases with ABL-class fusions, making it a specific marker for BCR-ABL1+ and BCR-ABL1-like B-lymphoblastic leukemia.
Furthermore, other studies have shown that positive MUC4 expression in breast cancer can lead to poorer prognosis and a poorer response to estrogen therapy; positive expression in ovarian cancer can serve as an early diagnostic indicator for epithelial ovarian cancer and is significantly correlated with tumor differentiation and lymph node metastasis; positive expression in colorectal cancer is an independent indicator of poor prognosis in early-stage colorectal cancer patients; positive expression in oral squamous cell carcinoma is an independent risk factor for poor prognosis; and expression in gastric cancer may become a new target for targeted therapy.
Extensive research indicates that MUC4 plays an important role in the biological processes of tumorigenesis and metastasis in many tumors and is a significant biomarker for multiple tumors:
-
It can be used for the early diagnosis of pancreatic precursor lesions and ductal adenocarcinoma, is also an independent factor for poor prognosis in pancreatic cancer patients, and can become a new target for targeted therapy in this tumor;
-
Its expression in non-small cell lung cancer can differentiate sarcomatoid mesothelioma from pulmonary sarcomatoid carcinoma and can become an important indicator for targeted therapy and immunotherapy in non-small cell lung cancer;
-
Its positive expression in endometrial cancer is higher than in adjacent tissues and is an important factor for poor prognosis in endometrial cancer;
-
It is a new diagnostic marker that can distinguish suspected meningioma from non-meningothelial tumors;
-
It is a specific marker for BCR-ABL1+ and BCR-ABL1-like B-lymphoblastic leukemia;
-
Its expression in multiple tumors such as breast cancer, ovarian cancer, colorectal cancer, oral squamous cell carcinoma, and gastric cancer has corresponding important roles in diagnosis, prognosis prediction, and treatment.
Related Antibodies from Maixin
|
Antibody Name
|
Product Number
|
Clone Number
|
Cellular Localization
|
|
MUC4
|
MAB-0749
|
8G7
|
Cytoplasm
|
References:
[1] Lin Xuan, Wang Hao, Cui Yunfu. Research Progress of Mucin-4 in Pancreatic Cancer[J]. Journal of Hepatopancreatobiliary Surgery, 2019, 31(11):695-700.
[2] Zhong Guodong, Lan Ting, Li Guoping, et al. Expression and Prognostic Significance of Mucins MUC1, MUC4 and MUC6 in Pancreatic Cancer[J]. Chinese Journal of Histochemistry and Cytochemistry, 2021, 30(6):515-524.
[3] Yonezawa S, Higashi M, Yamada N, et al. Precursor lesions of pancreatic cancer [J]. Gut Liver.
[4] Gautam S K , Kumar S , Cannon A , et al. MUC4 mucin- a therapeutic target for pancreatic ductal adenocarcinoma[J]. Expert Opinion on Therapeutic Targets, 2017.
[5] Zhu Peng, Liu Huiying, Jin Kaizhou, et al. Differential Expression Analysis of Mucin 4 in Pancreatic Intraepithelial Neoplasia and Pancreatic Cancer[J]. Chinese Clinical Oncology, 2014, 19(10):891-895.
[6] Zhou Zhuangzhuan, Lai Yanping. Research Progress on MUC4 Expression in Non-Small Cell Lung Cancer[J]. Jiangsu Medical Journal, 2021, 47(7): 739-743.
[7] Lee S R , Mun J Y , Jeong M S , et al. Thymoquinone-Induced Tristetraprolin Inhibits Tumor Growth and Metastasis through Destabilization of MUC4 mRNA[J]. International Journal of Molecular Sciences, 2019, 20(11):2614.
[8] Skg A , Sk A , Vd E , et al. MUCIN-4 (MUC4) is a novel tumor antigen in pancreatic cancer immunotherapy [J]. Seminars in Immunology, 47:101391
[9] Zhao Ying, Zhao Guangri, Wu Yunyu, et al. Study on the Expression of Disabled Homolog 2, Nectin-2, and Mucin 4 in Endometrial Cancer Tissues and Their Relationship with Prognosis[J]. Progress in Modern Biomedicine, 2021, 21(4):754-758.
[10] Shi Xiaozhe, Ma Hongfeng, Zhao Xianlan. Expression and Clinical Significance of Mucin 4 in Endometrial Cancer[J]. Journal of Practical Oncology, 2020, 35(1):65-68.
[11] Hasaneen Nermeen,Gad Allah Manal,Hewedi Iman,et al. Diagnostic role of MUC4 immunohistochemical expression in various subtypes of meningioma[J]. Egyptian Journal of Pathology,2020,40(2):243-248.
[12] Kaumeyer Benjamin,Fidai Shiraz,Sukhanova Madina,et al. MUC4 expression by immunohistochemistry is a specific marker for BCR-ABL1+ and BCR-ABL1-like B-lymphoblastic leukemia.[J]. Leukemia & lymphoma,2022.
[13] Dreyer Courtney A, VanderVorst Kacey, Free Savannah, et al. The role of membrane mucin MUC4 in breast cancer metastasis[J]. Endocrine-related cancer,2021.
For more information, please contact: 800-8581156 or 400-889-9853




