Epstein-Barr virus (EBV) is a specific lymphotropic herpesvirus in humans. Over 90% of the world’s population is infected with EBV, most of whom show no clinical symptoms and experience long-term latent infection. EBER is a small RNA encoded by EBV, discovered by Lerner et al., with a stable secondary structure. It exists in high copy numbers during all stages of EBV, including latency and replication.
EBV is both a causative factor for some non-neoplastic diseases (such as infectious mononucleosis) and one of the Group 1 carcinogens listed by the International Agency for Research on Cancer (IARC). It is closely associated with the development of various tumors, including lymphoma, nasopharyngeal carcinoma, gastric cancer, and post-transplant lymphoproliferative disorders.
EBV is closely related to the occurrence and development of various lymphomas, including Hodgkin lymphoma, Burkitt lymphoma, diffuse large B-cell lymphoma, angioimmunoblastic T-cell lymphoma, and NK/T-cell lymphoma. Currently, significant progress has been made in the research of EBV-associated lymphomas. Timely diagnosis and detection of EBV-related lymphomas can provide patients with more treatment opportunities and help improve the cure rate of the disease.
EBV-associated gastric cancer (EBVaGC) accounts for 6% to 12% of all gastric cancers and is one of the four molecular subtypes proposed by The Cancer Genome Atlas (TCGA) project. The immune microenvironment of EBVaGC is characterized by extensive lymphocyte infiltration, and patients are more sensitive to immunotherapy, making them a beneficiary group for immune checkpoint inhibitor therapy with a relatively favorable prognosis. Therefore, accurately diagnosing EBVaGC from gastric cancer is crucial for effective patient treatment.
Nasopharyngeal carcinoma (NPC) is a high-incidence tumor in southern China and is currently the most clearly defined human epithelial tumor associated with EBV infection. In the most common undifferentiated type of NPC, almost every cancer cell is infected with EBV. Increasing studies have shown that the detection of EBV has significant clinical implications for the early diagnosis of high-risk NPC patients, precise TNM staging, prediction of radiotherapy and chemotherapy responses, and prognosis assessment. It is an indispensable indicator in the diagnosis, treatment, and follow-up monitoring of NPC.
There are numerous methods for detecting EBV, but in tumor diagnosis, serological testing can only serve as an indication of EBV presence. Methods such as IHC detection of LMP are prone to false negatives due to the low expression of viral surface antigens during latency. PCR detection is complex and cannot precisely localize EBV. Leveraging the high copy number characteristic of EBER, EBER in situ hybridization can accurately locate the position and type of EBV-infected cells, providing reliable pathological evidence for clinical screening, diagnosis, treatment, and prognosis assessment of tumors. It is considered the ‘gold standard’ for pathological diagnosis of EBV.
Maixin EBER In Situ Hybridization Kit
The new EBER in situ hybridization kit, independently developed by the Maixin team, features optimally selected digoxigenin-labeled target-specific nucleic acid probes. The kit is fully equipped and can specifically bind to EBER, enabling precise localization of EBV-infected cells, effective signal amplification, high sensitivity, and strong specificity. The experimental process is flexible and controllable, with same-day completion possible (approximately 3 hours for manual staining; approximately 3.5 hours for Tian/Titan S machine staining). Overnight staining is also an option. It is suitable for EBV detection in paraffin sections of various tumor tissues. Additionally, to facilitate clinical experiments, the kit offers both manual and instrument-compatible formulations, allowing departments to flexibly choose based on actual needs. The probes in both formulations are ready-to-use, requiring no dilution before use, reducing operational errors and ensuring more accurate results and reliable data.
Staining Images Appreciation
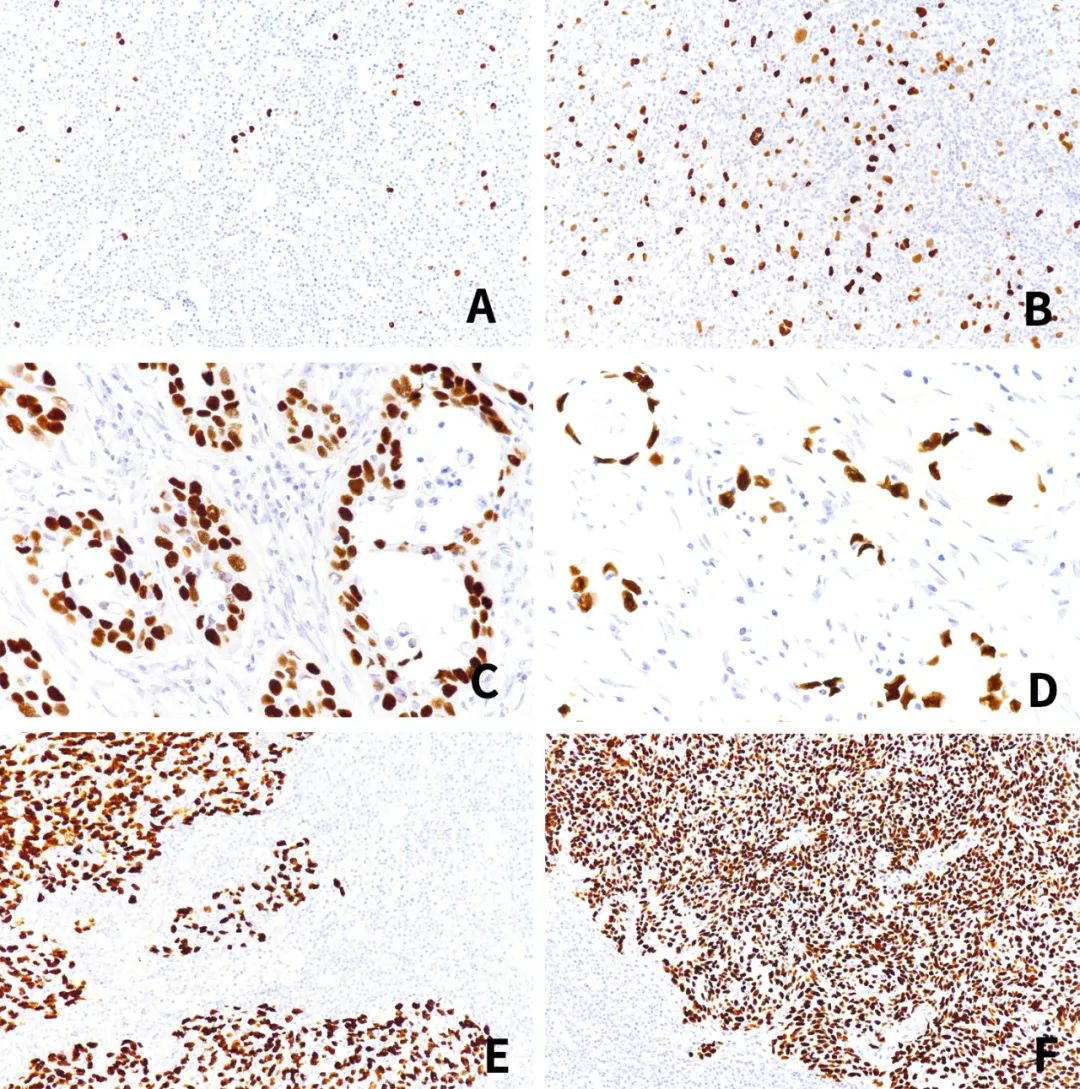
Figure:A、B:EBV-infected Hodgkin lymphoma tissue;C、D:EBV-infected gastric cancer tissue;E、F:EBV-infected nasopharyngeal carcinoma tissue.
References:
[1] National Children’s EBV Infection Collaborative Group, Editorial Board of Chinese Journal of Experimental and Clinical Virology. Expert Consensus on Laboratory Diagnosis and Clinical Application of EBV Infection [J]. Chinese Journal of Experimental and Clinical Virology, 2018, 32(1): 2-8.
[2]HualiYin, Jiani Qu, Qiu Peng, et al. Molecular mechanisms of EBV-driven cell cycleprogression and oncogenesis [J]. Medical Microbiology & Immunology, 2019, 208(5).573-583.
[3] Feng Jing, Wang Sanbin, Liu Lin, et al. Research Progress on the Pathogenesis of EBV Infection and Lymphoma [J]. Infectious Disease Information, 2022, 35(2): 172-175.
[4]The Cancer Genome Atlas Research Network. Comprehensive molecular characterization of gastric adenocarcinoma[J]. Nature.10.1038/nature13480
[5] Wu Ruoshi (Review), Nong Shengli (Reviewer). Research Progress on the Relationship Between EBV and the Development of Nasopharyngeal Carcinoma [J]. Modern Medicine & Health, 2016, 32(14): 2182-2184.
[6]Chua HueyHuey,Kameyama Toshiki,Mayeda Akila, et al.Epstein-Barr Virus Enhances Cancer-Specific Aberrant Splicing of TSG101 Pre-mRNA.[J]. International journal of molecular sciences,2022,23(5).
For more information, please contact: 800-8581156 or 400-889-9853


