Galectin-3 is a mammalian-derived lectin and glycoprotein component with various intracellular and extracellular functions, such as promoting tumor angiogenesis and growth, and facilitating tumor progression. Current studies indicate that galectin-3 can promote lymphocyte apoptosis, reduce the anti-tumor activity of NK cells, and inhibit T cell function through immune effects, particularly significantly affecting the function of CD8-positive T cells in HPV-positive head and neck squamous cell carcinoma. Literature also reports that galectin-3 expression is related to cancer progression; and related inhibitors have been approved for clinical studies in various cancers and other lesions.
To better understand the significance of galectin-3 in the progression and immune targeting of head and neck squamous cell carcinoma, pathologists Coppock et al. from the University of Virginia evaluated the relationship between galectin-3 expression and HR-HPV in primary head and neck squamous cell carcinoma and locally metastatic head and neck squamous cell carcinoma. The related article has been published in the journal “Head Neck Pathol”.
This study collected a total of 78 cases of primary and locally metastatic head and neck squamous cell carcinoma. After constructing tissue microarrays, HPV was assessed using HR-HPV RNA ISH, and galectin-3 expression was evaluated by immunohistochemistry. The results showed that among primary head and neck squamous cell carcinomas, 17 were HPV-positive and 36 were HPV-negative; among metastatic head and neck squamous cell carcinomas, 11 were HPV-positive and 14 were negative.
Immunohistochemical detection showed that the proportion of cases with tumor cell cytoplasmic positive expression ≥5% in all head and neck squamous cell carcinoma cases was 29% (23/78), as detailed in Table 1.
Table 1. Galectin-3 expression results under different conditions

Statistical analysis showed no significant difference in the proportion of galectin-3 positivity between primary and metastatic lesions (30%, 16/53; vs. 28%, 7/25; p=1). In primary HPV-positive head and neck squamous cell carcinomas, the galectin-3 positive proportion was 53% (9/17), while in metastatic HPV-positive head and neck squamous cell carcinomas, this positive proportion was 45% (5/11), with no significant difference between them (p=1). In primary HPV-negative head and neck squamous cell carcinomas, the galectin-3 positive proportion was 19% (7/36), while in metastatic HPV-negative head and neck squamous cell carcinomas, this positive proportion was 14% (2/14), with no significant difference between them (p=1).
However, when analyzing all HPV-positive cases together, the proportion of galectin-3 positivity was 50% (14/28), while in all HPV-negative cases, the proportion of galectin-3 positivity was only 18% (9/50), with a significant difference between them (p=0.004). Overall, HR-HPV positive cases are more likely to show galectin-3 positivity compared to negative cases.
Illustration Appreciation
Figure 1. HR-HPV in situ hybridization results:(A) HPV-positive squamous cell carcinoma;(B) HR-HPV RNA ISH detection, nuclear staining, cytoplasmic punctate staining;(C) HPV-negative squamous cell carcinoma;(D) HR-HPV RNA ISH detection negative.
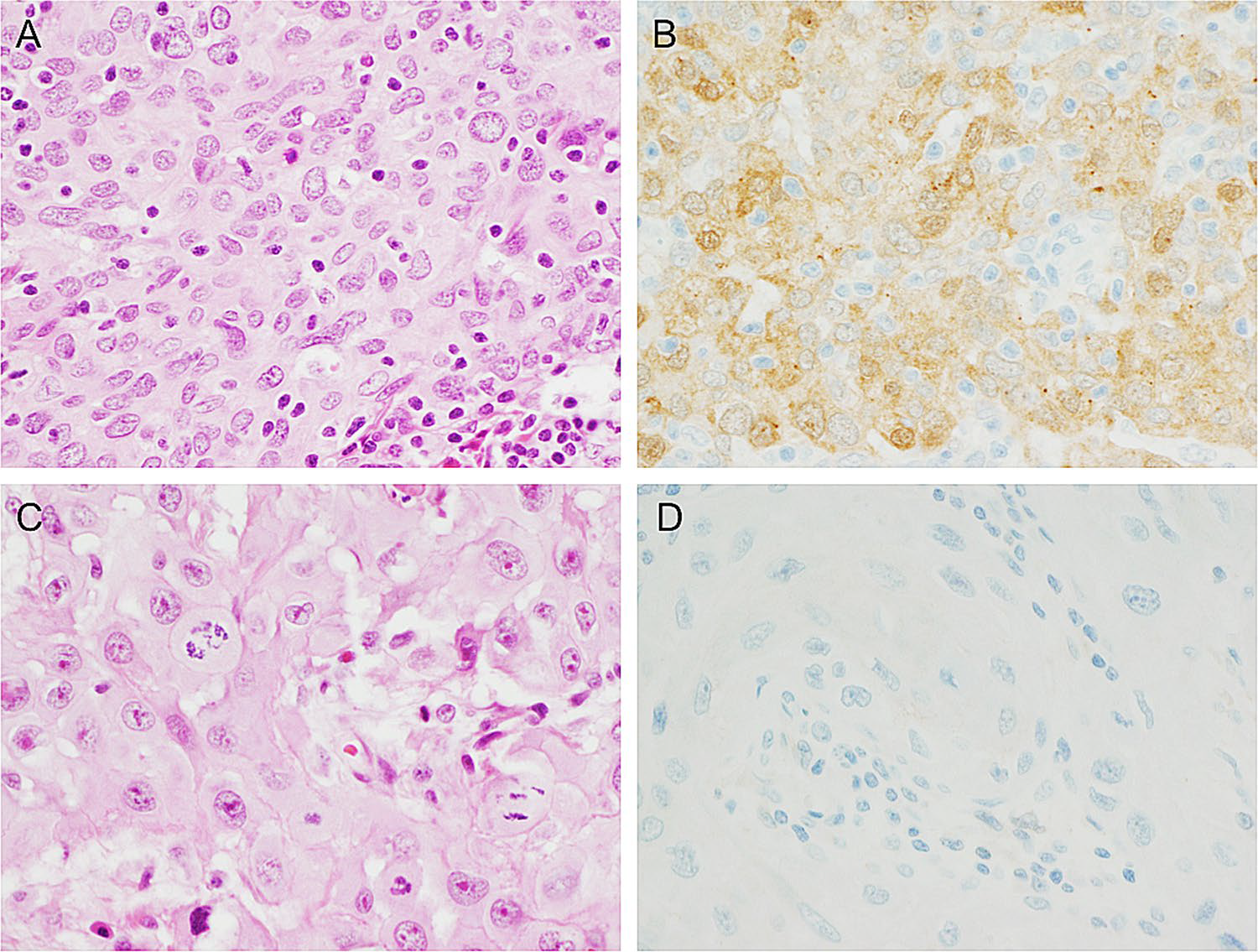
Figure 2. Immunohistochemical results of galectin-3 in primary head and neck squamous cell carcinoma: (A) galectin-3 positive oral primary squamous cell carcinoma; (B) immunohistochemical galectin-3 shows cytoplasmic staining in most tumor cells, with some nuclear staining; (C) galectin-3 negative oral primary squamous cell carcinoma for comparison; (D) complete absence of immunohistochemical galectin-3 staining.

Figure 3. Immunohistochemical results of galectin-3 in metastatic head and neck squamous cell carcinoma: (A) galectin-3 positive lymph node metastatic head and neck squamous cell carcinoma; (B) immunohistochemical galectin-3 shows positive staining in most tumors; (C) galectin-3 negative lymph node metastatic head and neck squamous cell carcinoma for comparison; (D) complete absence of immunohistochemical galectin-3 staining.
With the deepening of research on tumors and related lesions, galectin-3 inhibitors have gradually begun to be implemented in clinical trials. For example, GM-CT-01 has been approved for clinical trials in malignant melanoma (NCT01723813), and GR-MD-02 has been approved for studies in patients with liver fibrosis (NCT02462967) and for studies in combination with immune modulators and CTLA-4 inhibitors in metastatic malignant melanoma (NCT02117362). Although related data are limited, current research results indicate that galectin-3 antagonists are well tolerated in head and neck squamous cell carcinoma and may reduce the toxicity of other treatment regimens.
The results of this study show that galectin-3 is positively expressed in some head and neck squamous cell carcinoma cases, which may have certain implications for the implementation of galectin-3 inhibition therapy. In this study, HR-HPV positive head and neck squamous cell carcinomas had a significantly higher galectin-3 positive rate compared to HR-HPV negative head and neck squamous cell carcinomas, which may suggest that HPV infection plays some role in related lesions; and the lack of significant statistical difference in galectin-3 expression between metastatic and primary lesions in this study does not support that galectin-3 expression promotes metastatic progression.
|
Antibody Name
|
Product Number
|
Clone Number
|
Positive Site
|
|
Galectin-3*
|
MAB-0835
|
MX060
|
Cytoplasm/Nucleus
|
*Marked asMaxin Clone Product
Coppock JD, Mills AM, Stelow EB. Galectin-3 Expression in High-Risk HPV-Positive and Negative Head & Neck Squamous Cell Carcinomas and Regional Lymph Node Metastases [published online ahead of print, 2020 Jun 20]. Head Neck Pathol. 2020;10.1007/s12105-020-01195-3. doi:10.1007/s12105-020-01195-3




