FoxP3 (forkhead box Protein 3) is the master transcription factor for regulatory T cell (Treg) lineage. FoxP3 plays a crucial role in Treg cell development and immunoregulatory functions. The absence of FoxP3 in Treg cells can confer pro-inflammatory properties, thereby exacerbating the progression of autoimmune diseases or significantly enhancing immunosuppressive effects. A growing body of research confirms that FoxP3 is not only specifically expressed in thymic or peripherally derived Treg cells but is also aberrantly expressed in various tumor cells, closely related to tumorigenesis, development, and prognosis.
The expression and functions of FoxP3 vary significantly across different tumors. When acting as a tumor suppressor gene, it can regulate the expression of downstream oncogenes and other signaling pathways to inhibit tumor initiation and development. However, in some tumors, its expression endows tumor cells with Treg-like functions, linking it to tumor progression and immune escape, thereby playing a pro-tumorigenic role.
Treg cells are crucial for regulating immune responses and maintaining immune homeostasis in the body, and they also pose an obstacle to the efficacy of tumor immunotherapy. Studies have found that Treg cells with high FoxP3 expression exhibit significant immunosuppressive effects. They suppress the activation and proliferation of various immune cells, such as CD4+ Th1 cells, through direct contact via surface membrane molecules or by secreting inhibitory cytokines like IL-10 and TGF-β. The role of FoxP3 is primarily manifested in the following aspects: ① Removal of Treg cells or inhibition of their function can upregulate anti-tumor immune responses; ② Downregulation of FoxP3 expression modulates the immunosuppressive function of T cells, enhancing immune responses; ③ Use of anti-cytokine monoclonal antibodies to block the immunosuppressive function of regulatory T cells.
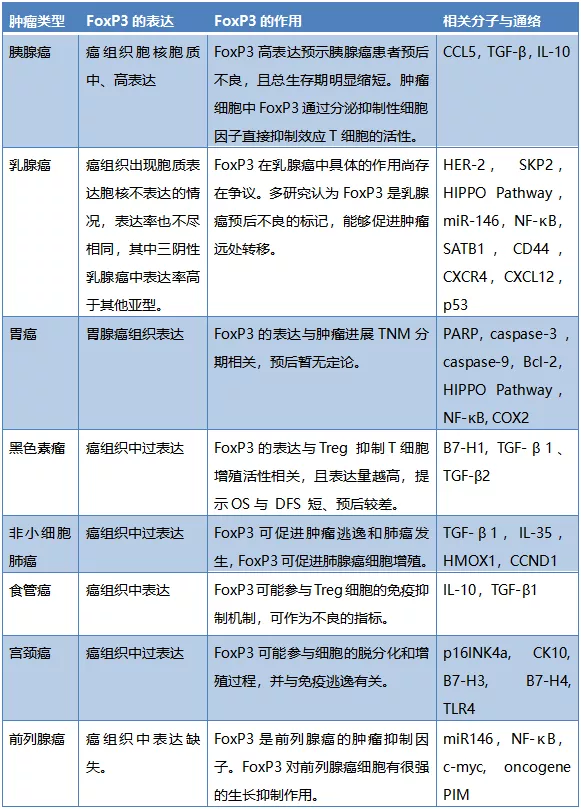
Applications of FoxP3 in Different Tumors
Li Xin et al. collected 120 surgically resected pancreatic cancer samples from Tianjin Medical University Cancer Hospital. Immunohistochemical staining revealed that FoxP3 expression was negative in normal pancreatic tissues and low-grade malignant pancreatic tumors (including serous cystadenoma, pancreatic intraepithelial neoplasia, and neuroendocrine tumors). However, in pancreatic ductal adenocarcinoma and four pancreatic cancer cell lines (PANC-1, BxPC-3, ASPC-1, MIA PaCa-2), FoxP3 was mostly moderately to highly expressed in both the nucleus and cytoplasm. The strong positive group showed a significant correlation with malignant phenotypes such as tumor size and shorter survival, suggesting poor prognosis for patients with high FoxP3 expression.
FoxP3 expression is negative in normal cervical epithelium, while the proportion of FoxP3+ T cells increases in cancerous tissues, playing a significant role in the occurrence and development of cervical cancer.In clinicopathological features, FoxP3 expression is significantly correlated with FIGO stage and tumor size.Simultaneously, an increase in Treg numbers is positively correlated with the degree of lymph node and hematogenous metastasis in cervical cancer.Additionally, the correlation between FoxP3 and B7-H3, B7-H4, and TLR4 indicates its important role in the tumor immune escape process.Therefore, FoxP3 and Treg have become new targets for clinical treatment of cervical cancer.
Li Min et al. detected the expression of FoxP3 protein in esophageal cancer tissues using immunohistochemistry and analyzed its clinical significance. The results showed that FoxP3 protein was mainly expressed in the nuclei of tumor-infiltrating lymphocytes and tumor cells, with no expression in adjacent tissues. Among patients with high FoxP3 expression in tumor cells (H-score ≥ 70), the proportion of patients with tumor size ≥ 3.5 cm was significantly higher than in patients with low FoxP3 expression in tumor cells (H-score < 70), with a statistically significant difference. However, there was no statistically significant difference in FoxP3 expression levels in tumor cells with other clinical parameters. The conclusion was: FoxP3 expression in esophageal cancer tissues is mainly distributed in infiltrating lymphocytes and tumor cells. FoxP3 expression is significant in tumor progression and can serve as a potential prognostic indicator for clinical diagnosis and treatment of esophageal cancer.
Research by Wang Zhonglin and Xiang Rengyun et al. showed that FoxP3 is expressed in gastric cancer cells at higher levels than in normal gastric mucosa tissues. The expression level of FoxP3 in gastric cancer tissues was not correlated with patient age, gender, tumor differentiation, tumor size, or lymph node metastasis, but was significantly correlated with tumor TNM stage. This indicates that FoxP3 is related to the development and malignancy of gastric cancer to some extent, but not to patient prognosis. Research by Lu Changqing showed that FoxP3 expression is related to lymph node metastasis, and patients with high FoxP3 expression have a worse prognosis.
Studies show that FoxP3 is highly expressed in normal breast epithelium but significantly reduced in breast cancer tissues. Zuo et al. found through detection of human and murine tumor tissues and breast cancer cell lines that FoxP3 expression at both transcriptional and tissue levels was lower to varying degrees compared to normal breast epithelial tissues. In other studies, higher FoxP3 expression in breast cancer was associated with lower patient survival rates, and FoxP3 positivity could serve as a molecular marker for poor prognosis.
FoxP3 is lowly expressed in normal bronchial epithelium but overexpressed in tumor tissues and adjacent tissues. It is expressed in both the nucleus and cytoplasm of non-small cell lung cancer cells, and its expression is significantly correlated with lymph node metastasis and TNM stage. Fu et al. found that FoxP3 and TLR4 expression were both high in non-small cell lung cancer tissues and cell lines and were positively correlated. Li et al. also found that FoxP3 expression was positively correlated with the expression of immunosuppressive factors TGF-β1, IL-35, and HMOX1, suggesting FoxP3 involvement in the immune escape state of non-small cell lung cancer. On the other hand, overexpression of FoxP3 promotes tumor cell proliferation, migration, and invasion. When FoxP3 expression is specifically reduced, the corresponding malignant biological behaviors also decrease. In-depth mechanistic studies found that FoxP3 expression is significantly correlated with the expression of the cell cycle G1/S checkpoint gene CCND1. FoxP3 can increase CCND1 expression, thereby promoting the proliferation of non-small cell lung cancer cells. The above research results indicate that FoxP3 plays a pro-tumorigenic role in lung cancer.
Table 1: Expression, Function, and Related Molecules of FoxP3 in Different Tumors
(Click to view larger image)
Research on FoxP3 in the aforementioned different tumors reveals that FoxP3 regulates the expression of various downstream genes in both Treg cells and tumor cells, exerting multifaceted effects. It directly influences the initiation, development, proliferation, apoptosis, invasion, and migration of tumor cells; participates in remodeling the tumor immune microenvironment, tumor immune escape, and immunosuppressive states; and by assessing FoxP3 expression in tumor cells, it can reflect the immune status in the microenvironment and indicate prognosis. However, the expression and significance of FoxP3 in tumors remain controversial. As a new immune response indicator, its specific mechanisms require further in-depth study.
Related Antibodies from Maixin
|
Antibody Name
|
Product Number
|
Clone Number
|
Cellular Localization
|
|
FoxP3
|
MAB-1004
|
236A/E7
|
Nucleus
|
References:
[1] Ren He, Li Xin. Research progress on the expression and function of transcription factor FoxP3 in different tumors[J]. Chinese Journal of Clinical Oncology, 2017, 44(10): 508-514.
[2] Li Min, Chen Lujun, Jiang Jinting. Expression and clinical significance of nuclear transcription factor FoxP3 in esophageal cancer cells[C]// National Immunology Conference. 2014.
[3] Wang Jia, Fan Xiaoyan, Sha Min, et al. FoxP3 and cervical cancer [J]. International Journal of Immunology, 2017, 40 (01): 107-112. DOI: 10.3760/cma.j.issn.1673-4394.2017.01.023
[4] Guo Jing, Wang Juyong, Zheng Zhan. Transcription factor FoxP3 and lung cancer [J]. International Journal of Oncology, 2011, 38(09): 677-679. DOI: 10.3760/cma.j.issn.1673-422x.2011.09.011
[5] Wang Zhonglin, Qiu Lei, Feng Chuanbo, et al. The impact of FoxP3 expression in gastric cancer tissues on clinical stage and prognosis [J]. International Journal of Surgery, 2020, 47 (08): 522-526, f3. DOI: 10.3760/cma.j.cn115396-20200424-00113
[6] Xiang Rengyun, Li Kai, Xiang Ping. Expression of FoxP3 in gastric cancer cells and its clinical significance [J]. Journal of Chinese Physicians, 2017, 19 (05): 712-720. DOI: 10.3760/cma.j.issn.1008-1372.2017.05.020
[7] Li Jingping, Zhang Xiangmei, Zheng Lihua, Liu Yunjiang. Research progress on the expression of FoxP3 in breast cancer cells[J]. Cancer Research on Prevention and Treatment, 2016, 43(12): 1076-1080.


