MaiXin’s Self-Developed Novel Liver Cancer Detection Kit, in Collaboration with Academician Wang Hongyang’s Team, Receives First CFDA Certification.
In August 2014, Fuzhou Maixin Biotechnology Development Co., Ltd. (hereinafter referred to as “Maixin Company”) collaborated with Academician Wang Hongyang and her team from the Surgical Research Institute of Shanghai Eastern Hepatobiliary Surgery Hospital to jointly develop the Glypican-3 (GPC3) detection kit (immunohistochemistry method), which obtained the Class III in vitro diagnostic immunohistochemistry product registration certificate issued by the China Food and Drug Administration (CFDA).


As is well known, liver cancer is one of the most common malignant tumors in humans. Clinically, alpha-fetoprotein (AFP) testing and B-ultrasound examinations are often performed every six months to achieve early diagnosis and monitoring of liver cancer. However, practical experience shows that imaging examinations are not sensitive to early liver lesions and rely on the operator’s experience, presenting significant limitations. Although serum AFP testing is widely recognized as the first choice for screening due to its simplicity, low invasiveness, and low cost, the positive detection rate of AFP is less than 50%. Moreover, a considerable number of chronic hepatitis and cirrhosis cases show positive serum AFP levels, resulting in certain rates of missed and misdiagnosis. Therefore, there is an urgent need to find new liver cancer markers.
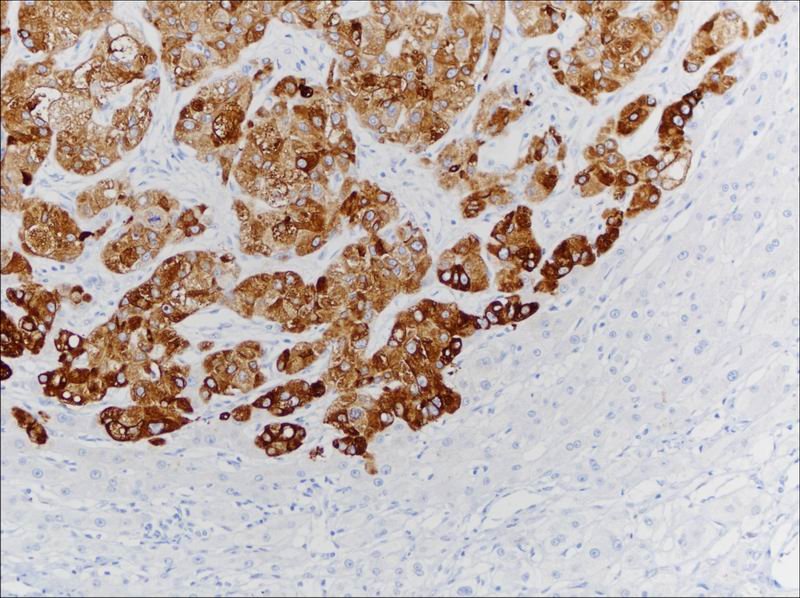
Academician Wang Hongyang is an outstanding female leader in the field of biological signal research. As one of the youngest female academicians in China, she established the country’s first international cooperative biological signal transduction research center as early as 1997. Bearing the mission of solving the liver cancer problem as soon as possible, she pioneered the discovery of new liver cancer diagnostic markers internationally. Overcoming the immaturity of monoclonal antibody preparation technology in the early stages of research with her unique perseverance, she achieved breakthrough progress and fruitful results. GPC3 is one of them. Research indicates that GPC3 plays an important role in the development of primary liver cancer and can serve as a new marker for primary liver cancer, especially for AFP-negative liver cancer. The classic liver cancer marker AFP has low sensitivity and low expression rate in liver cancer tissues, with a positive expression rate of about 30%. In contrast, the positive expression rate of GPC3 in liver cancer can reach 80%. GPC3 has high sensitivity and specificity, with high expression in hepatocellular carcinoma and no or low expression in benign liver lesions and precancerous lesions. It helps differentiate small liver cancer from high-grade dysplasia. Performing GPC3 immunohistochemical staining on liver biopsy specimens can serve as an effective auxiliary tool for early differential diagnosis of liver cancer, aiding in predicting treatment and prognosis improvement for HCC patients.
According to the agreement signed by both parties, Maixin Company provides one-stop technical support for the research and development optimization, clinical trials, mass production, and registration application of the GPC3 detection kit. For 20 years, Maixin has been committed to providing high-quality, comprehensive pathological solutions for pathology departments and has gradually formed a complete R&D platform covering the upstream, midstream, and downstream of immunohistochemistry. To change the current production and usage status of domestic immunohistochemistry products, the company has invested heavily, actively responded to relevant national policy requirements, strictly controlled product quality, and persevered through challenges. It has become one of the earliest domestic reagent manufacturers to obtain the CFDA Class III immunohistochemistry registration certificate and remains one of the enterprises with the most CFDA Class III tumor-related detection reagent registration certificates. It is believed that this industry-research collaboration will help both parties establish a long-term mutual trust cooperation mechanism, advance liver cancer diagnosis and treatment research, assist in the industrial production and clinical application of domestic antibodies, and thereby establish a technological innovation platform with independent intellectual property rights in the field of in vitro diagnostic reagents. This also holds significant strategic importance for breaking the long-term monopoly of foreign enterprises and increasing the market share of domestic in vitro diagnostic reagents in the high-end healthcare market.

Glypican-3 staining in primary hepatocellular carcinoma, cytoplasmic positive

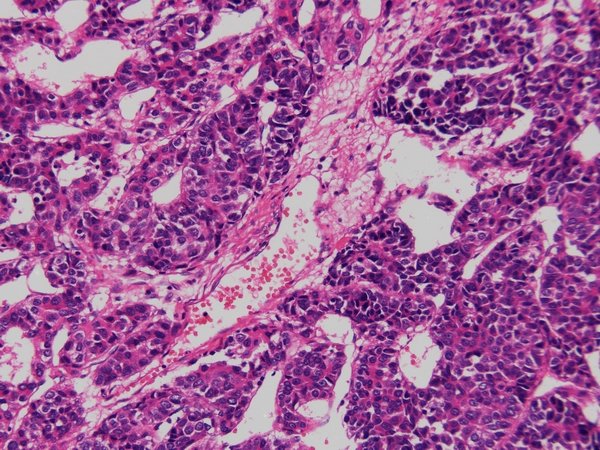
HE staining

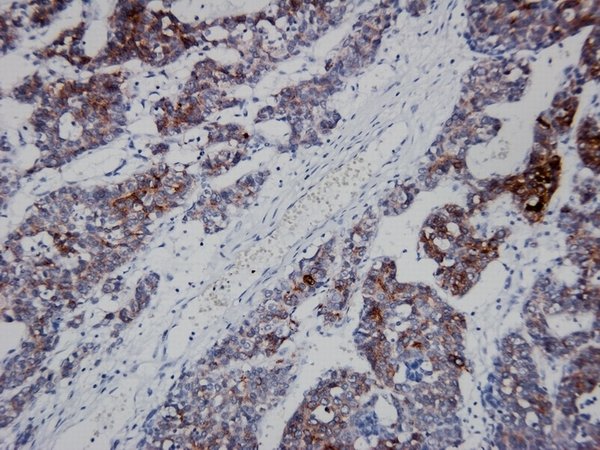
GPC-3 staining
Product Information:
| Product Number |
Clone Number |
Registration Certificate Number |
| MAB-0667 | MAXIM001 | National Medical Device (Approval) No. 2014 3401502 |
Further Reading:Science and Technology Daily – China Science and Technology Network http://www.wokeji.com/shouye/zxfxycx/201409/t20140927_828365.shtml
