The diagnosis of renal cell carcinoma (RCC) typically requires careful histological examination and a limited immunohistochemistry (IHC) panel. Over 90% of adult renal tumors are clear cell, papillary, chromophobe RCC, and oncocytoma, making complex ancillary tools often unnecessary. Renal tumors with solidly defined genetic alterations may ultimately require molecular confirmation through cytogenetic or sequencing techniques, such as RCC with TFE3, TFEB, or ALK gene rearrangements, or RCC with TFEB amplification. In fumarate hydratase-deficient and succinate dehydrogenase-deficient renal cell carcinomas, highly specific IHC markers can strongly suggest the diagnosis. In metastatic cases, PAX8 and CA IX are the most helpful markers for confirming RCC and clear cell type, respectively; however, caution should be exercised in the absence of a current or historical renal mass. In diagnostically challenging cases, careful examination combined with judicious IHC markers usually resolves the diagnosis. This review provides a concise overview of daily pathological practice in diagnosing renal tumors with the latest confirmed, provisional, and emerging entities.
Over the past decade, the number of newly confirmed distinct renal entities has rapidly expanded, with approximately 90% of adult renal tumors being clear cell RCC (CCRCC), type 1 papillary RCC/papillary adenoma, chromophobe RCC, and oncocytoma. Renal tumors often exhibit distinctly recognizable cellular morphology, and the currently used limited IHC is usually sufficient for accurate diagnosis. Some renal tumors with solidly defined molecular alterations have sensitive and specific IHC tests, particularly fumarate hydratase-deficient RCC (FH-RCC), succinate dehydrogenase-deficient renal cell carcinoma (SDH-RCC), and TFE3-rearranged RCC. In this review, we outline the judicious use of immunohistochemistry in identifying renal tumors, grouping renal tumors according to their prominent cellular morphological features.
Role of Renal Cell Carcinoma Biopsy
In the management of suspected renal masses, percutaneous core needle biopsy has been increasingly utilized as a minimally invasive diagnostic and potentially therapeutic tool (e.g., ablation). Renal mass biopsy (Figure 1A) provides an accurate diagnosis.
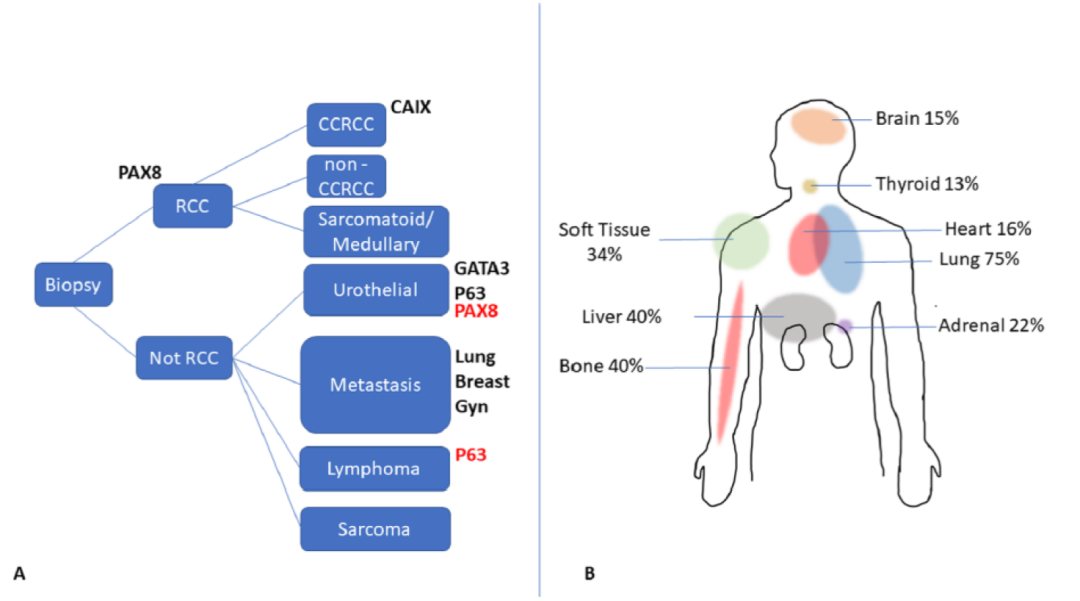
Figure 1. Algorithm for renal core needle biopsy (A) and metastatic sites (B). Positive markers in red can be deceptive, such as PAX8 positivity in urothelial carcinoma or p63 positivity in lymphoma.
In a recent meta-analysis, the overall median diagnostic rate was 92%, with a sensitivity of 99.1% and specificity of 99.7%. On the other hand, 16% of renal cancers develop distant metastases, with the lungs, bones, liver, and soft tissues being the most common sites (Figure 1B). However, renal cell carcinoma can metastasize to unusual sites, including but not limited to the skin, oral cavity, parotid gland, breast, or female genital tract. Clinical history is crucial when evaluating metastatic renal cell carcinoma. The incidence of metastatic progression in clinically localized renal masses (cT1a-cT1b) is as high as 6%. However, metastatic renal cell carcinoma is unlikely in the absence of a known primary renal tumor, so caution should be exercised.
PAX8 is a transcription factor composed of 450 amino acids (~48 kDa), involved in the embryogenesis of various organs, often positive in renal tubules, collecting ducts, and Bowman’s capsule parietal cells, with lesser positivity in adult renal pelvic urothelium. Most primary renal tumors are PAX8 positive (Figure 2). Similar positivity rates have been found in collecting duct carcinoma (CDC), renal medullary carcinoma (RMC), mucinous tubular and spindle cell carcinoma (MTSCC), metanephric adenoma, acquired cystic kidney disease-associated RCC (ACKD-RCC), mixed epithelial and stromal tumor (MEST), and unclassified RCC. Sarcomatoid dedifferentiation in RCC is associated with reduced PAX8 labeling, but PAX8 usually remains positive. Yu et al. reported that 27/42 (64%) sarcomatoid dedifferentiated RCCs had PAX8 labeling, and another study by Suster et al. showed a similar PAX8 expression rate (30/49, 61%). On the surface, PAX8 is a sensitive and specific marker for metastatic RCC, reliably used to differentiate metastatic clear cell RCC (83%-100%), papillary RCC (100%), and chromophobe RCC (57%-100%).
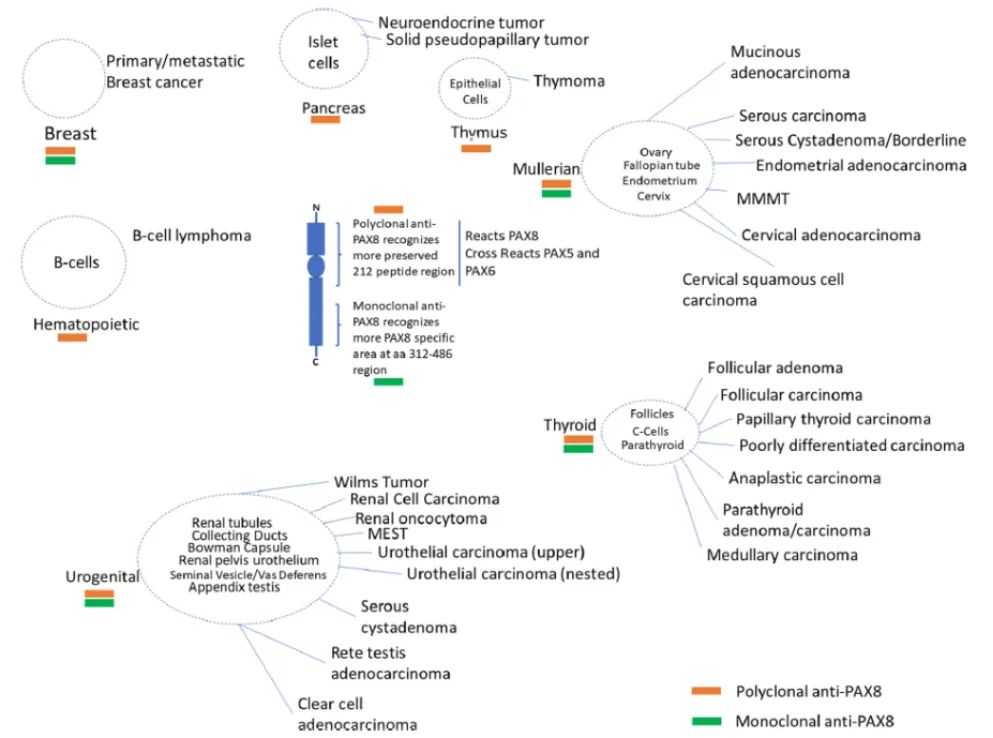
Figure 2. PAX8 protein structure, monoclonal and polyclonal antibodies, PAX8 expression in normal and tumor tissues. MEST: mixed epithelial and stromal tumor; MMMT, malignant mixed müllerian tumor.
PAX8 positivity has been reported in some upper urinary tract urothelial carcinomas (7%-17%), as well as in some urothelial carcinomas originating from the bladder, particularly nested variant urothelial carcinoma. In high-grade tumors involving the renal medulla/renal sinus/renal pelvis, PAX8, p63, and GATA3 are usually helpful in distinguishing RCC from urothelial carcinoma. The anti-PAX8 antibody clone is also important, especially when evaluating metastatic lesions. Most studies evaluating PAX8 expression in renal tumors have used rabbit polyclonal PAX8, which binds to the N-terminal portion common to all PAX family proteins. In contrast, using monoclonal PAX8 antibodies appears to increase specificity for detecting renal and ovarian cancers without labeling B lymphocytes and islet cells. However, a recent interesting study found that primary (8/27, 30%) and metastatic (10/22, 45%) breast cancers were labeled with monoclonal PAX8 (mostly focal and weak to moderate intensity), while no primary breast cancers expressed rabbit polyclonal PAX8.
Another renal lineage marker, PAX2, which closely collaborates with PAX8 in renal embryogenesis, has similar expression in RCC but may be less reliable in high-grade tumors and is less commonly used in uropathology practice. Ozcan et al. reported PAX2 expression rates of 92%, 87%, 83%, 100%, and 88% in clear cell RCC, papillary RCC, chromophobe RCC, collecting duct RCC, and oncocytoma, respectively, although PAX2 was not expressed in the sarcomatoid components of RCC. Most metastatic RCCs (74%-100%) have been reported to express PAX2.
Two other markers sometimes used are CD10 and RCC marker/antigen. CD10 is expressed by renal tubular cells (brush border) and by RCCs derived from proximal tubules, as well as by non-renal tumors, including urothelial, prostate, and pancreatic adenocarcinomas; endometrial stromal sarcoma or melanoma. CD10 may be helpful in some cases for RCC subtyping, but it is not specific to renal origin. Similarly, the RCC marker is typically positive in RCCs thought to originate from proximal tubules, although it is also positive in some non-renal tumors, including breast, prostate, and colon adenocarcinomas; melanoma, adrenal cortical, and parathyroid tumors.



