Immunohistochemical Antibody Selection Scheme for Spleen Diseases (Part 1)
Introduction:
Currently, immunohistochemistry has become a routine tool in pathological differential diagnosis, but the interpretation of immunohistochemistry in splenic diseases remains confusing. This article introduces the latest knowledge on the immunophenotypes of normal spleen and benign and malignant splenic diseases. Due to space limitations, it will be published in five parts, hoping to be helpful to colleagues in their daily work.
Histology of the Spleen
To correctly interpret immunohistochemical results of the spleen, we first need to understand the histology and function of the normal spleen. This also helps us determine which part has changed when assessing splenic lesions.
|
Basic Spleen Immunohistochemistry Panel: CD3, CD8, CD20,CD34, ERG, WT1 |
|
Red Pulp Lymphoid Lesions: CD8, CD68, CD34, DBA.44, annexin A1, BRAF V600E,CD25, CD103 |
|
White Pulp Lymphoid Lesions: CD20, CD3, CD5, CD10, BCL6, BCL1 (cyclin D1), BCL2,Ki-67, MUM1, EBER, CD30, CD8 |
|
Vascular Lesions: ERG, CD31, CD34, factor VIII, D2-40, CD68, CD163, CD8,WT1 |
|
Stromal/Histiocytic Lesions: CD21, CD23, CD35, LMP1, EBER, SMA, S100, ERG, CD31,CD34, WT1, CD68, CD163, CD1a ,CD3 |
|
TCell orNKCell Lymphoma/Leukemia: CD2, CD3, CD5, CD7, EBER, TIA1, granzyme B, TCL1,CD56, CD4, CD8, CD30 |
|
Cysts: CD8, cytokeratin AE1/AE3, calretinin, GMS ERG |
Table 2. Comparison of Vascular Markers in the Spleen
|
Antibody |
Specificity |
Sensitivity |
Characteristics |
|
ERG |
↑↑↑ | ↑↑↑↑ |
Nuclear positive, marks vascular endothelial and vascular tumors |
|
WT1 |
↑↑↑ | ↑↑↑ |
Cytoplasmic positive, background staining isCD34light |
|
CD34 |
↑↑↑ | ↑↑ |
Cannot mark splenic littoral cells, splenic hamartoma, or littoral cell angioma |
|
CD31 |
↑↑ | ↑↑↑ |
Non-specific, also marks histiocytes and macrophages |
|
VIII Factor |
↑↑ | ↑↑ |
Second-line antibody, less used, moderate sensitivity and specificity |
↑↑Indicates lower specificity and/or sensitivity;↑↑↑Indicates moderate specificity and/or sensitivity;↑↑↑↑Indicates highest specificity and/or sensitivity.
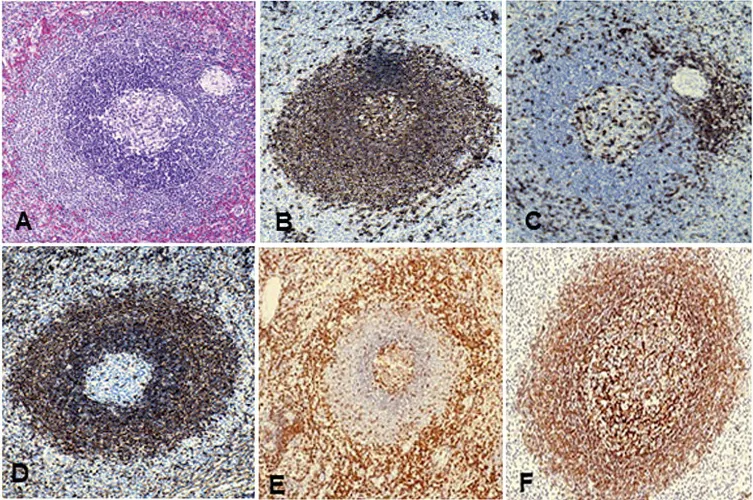
Figure1 Immunophenotype of normal spleen.
Figure1A, white pulp, showing germinal center and mantle zone/Marginal zone;
Figure1B,CD20showing white pulpBcell positive;
Figure 1C, CD3 is expressed in scattered T cells of the white pulp.
Figure1D,Bcl-2Shows negative in germinal centers of normal white pulp, but usually marginal zone/mantle zone positive;
Figure1E,CD43Expressed inTcells of normal white pulp;
Figure1F,CD21Highlights the follicular dendritic reticular structure in the white pulp;
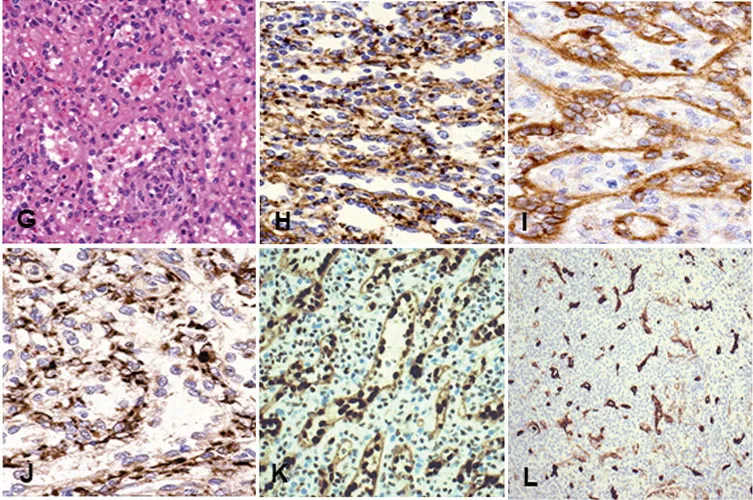
Figure1GtoM, red pulp;
Figure1G, red pulp showing splenic sinuses;
Figure1H,VIIIFactor stains endothelial cells;
Figure1I,CD8Highlights the splenic sinus structure;
Figure1J,CD68Shows positive in histiocytes;
Figure1K,ERGMarks endothelial cells;
Figure1L,CD34Also marks endothelial cells;
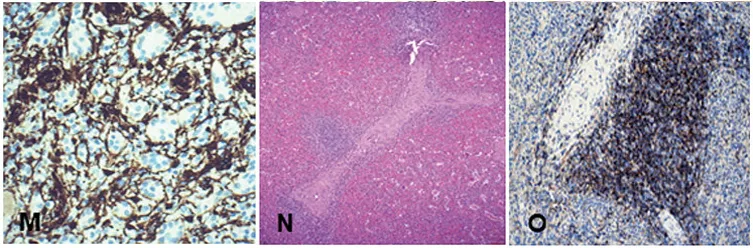
Figure1M,WT1is a very sensitive endothelial marker;
Figure1NandO, showingTzone.N, periarteriolar lymphoid sheath, surrounded by lymphocytes;
Figure1O,CD4Shows helperTcells.
Flow Cytometry
When performing flow cytometry analysis on normal splenic tissue, some abnormal lymphocyte subsets may be encountered. Approximately20% to30% of non-neoplastic splenicBlymphocytes expressCD5. In human immunodeficiency virus (HIV) and autoimmune diseases, the number of theseCD5-positive non-neoplasticBlymphocytes can increase. These non-neoplasticBcells should not be confused with mantle cell lymphoma or chronic lymphocytic leukemia//small lymphocytic lymphoma, both of which expressCD5. A small number ofCD4 +/ CD8+Tcells are usually present in normal spleen, with an average number accounting forT% of3.3cells, up to6%; usuallyCD4fluorescencestaining is bright, whileCD8fluorescencestaining is dim, and should not be misdiagnosed asTcell lymphoblastic leukemia//lymphoma. SomeTcell antigens may be lost in splenicTcells, so they should not be considered neoplastic. A portion of normalTlymphocytes in the spleen do not expressCD7. The proportion ofγδTcells in the spleen (12.5%±8.1%) is higher than in peripheral blood (4.0%±3.1%); theseγδTcells usually expressCD3, but notCD4,CD8andCD5. These findings are related to neoplasia in theαβTcell population, notγδTcells.Studies have shownthat approximately5%of splenicTcells areCD5 -; most of these cells areCD8+ or areγδTcells.
The number of immature phenotype (NK-) natural killer (CD56+ / CD16 ) cells in the spleen is higher than in peripheral blood. Additionally,CD2–NKcells constitute an important normal cell subset in splenic tissue. AlthoughCD7expression is considered a characteristic ofNKcells, it is common in the spleen.CD7expression is widespread.
Although flow cytometry is an excellent method for immunophenotyping, such as in lymphomas, for splenic lesions (such as vascular hyperplasia or stromal lesions), histological section examination is essential to assess splenic structure and immunohistochemical expression.
To be continued.…..
Maxin Related Antibodies
|
Antibody Name |
Product Number |
Clone Number |
Positive Location |
|
CD3* |
MAB-0740 |
MX036 |
Cell Membrane |
|
CD3 |
Kit-0003 |
SP7 |
Cell Membrane |
|
CD8 |
RMA-0514 |
SP16 |
Cell Membrane |
| CD8 |
MAB-0021 |
C8/144B |
Cell Membrane |
|
CD20 |
Kit-0001 | L26 | Cell Membrane |
| CD34 | Kit-0004 | QBEnd/10 |
Cell Membrane/Cytoplasm |
| ERG* |
RMA-0748 |
MXR004 | Nucleus |
| WT1* |
MAB-0678 |
MX012 |
Nucleus |
*Marked as Maxin clone products
