Great News! | Maxin’s Fully Automated Immunohistochemistry Stainer Secures Class I Medical Device Production Filing Certificate!
Introduction:
As early as 2016, the State Council issued the “13th Five-Year National Science and Technology Innovation Plan” (State Council Document [2016] No. 43), which clearly outlined the focus on localization in the in vitro diagnostic product sector, specifically mentioning the development of major products such as rapid pathological diagnosis systems,to enhance the competitiveness of China’s in vitro diagnostic industry.
In recent years, national and provincial-level authorities have repeatedly issued documents and formulated a series of measures to encourage the localization of medical devices, throughcore technology research and development, breaking foreign monopolies, and gradually changing the industry’s situation of being constrained by others with“domesticsubstitution”Form.

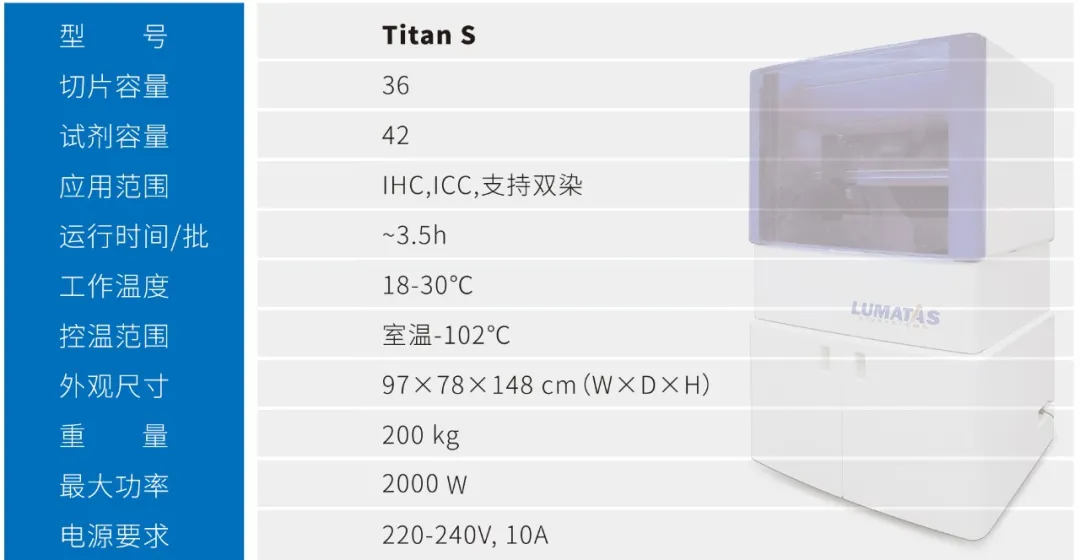
About Titan S
Titan S Core Advantages
-
High system sensitivity
-
Broad primary antibody compatibility
-
Stable results
-
Powerful functionality
-
High cost-effectiveness
-
Simple operation
-
After-sales support
Bearing the Mission
