Solid-Nested/Clear-Spindle
This category of tumors [Table 1] exhibits solid to nested growth, with cell morphology ranging from epithelioid to plump spindle-shaped, occasionally showing clear cytoplasmic changes. This category includes myoepithelial carcinoma (MyoCA), epithelial-myoepithelial carcinoma (EMC), clear cell carcinoma (CCC), and squamous cell carcinoma (SqCC). MyoCA consists of a single cell population with a myoepithelial phenotype, appearing as nondescript rounded spindle to round cells within a myxoid stroma. EMC is biphasic, composed of plump spindle to polygonal myoepithelial cells interspersed with tubular structures lined by epithelial cells. Both can show multilobular to nodular growth with central necrosis, and myoepithelial cells may have clear or non-clear cytoplasm. Salivary CCC contains nests or cords of cells with clear or pale cytoplasm, pyknotic nuclei, and dense eosinophilic hyalinized stromal bands. SqCC is a single cell population with dense, eosinophilic cytoplasm and frequent mitoses. Prominent keratinization may sometimes occur, but not always. Primary parotid squamous cell carcinoma remains controversial; any squamous cell carcinoma involving a major salivary gland should be considered as metastasis from head and neck skin or direct extension from mucosal sites.bright cytoplasmic changes. This category includes myoepithelial carcinoma (MyoCA), epithelial-myoepithelial carcinoma (EMC), clear cell carcinoma (CCC), and squamous cell carcinoma (SqCC). MyoCA consists of a single cell population with a myoepithelial phenotype, appearing as nondescript rounded spindle to round cells within a myxoid stroma. EMC is biphasic, composed of plump spindle to polygonal myoepithelial cells interspersed with tubular structures lined by epithelial cells. Both can show multilobular to nodular growth with central necrosis, and myoepithelial cells may have clear or non-clear cytoplasm. Salivary CCC contains nests or cords of cells with clear or pale cytoplasm, pyknotic nuclei, and dense eosinophilic hyalinized stromal bands. SqCC is a single cell population with dense, eosinophilic cytoplasm and frequent mitoses. Prominent keratinization may sometimes occur, but not always. Primary parotid squamous cell carcinoma remains controversial; any squamous cell carcinoma involving a major salivary gland should be considered as metastasis from head and neck skin or direct extension from mucosal sites.
Table 1. Immunophenotype and genetics of tumors exhibiting solid-nested/clear-spindle patterns
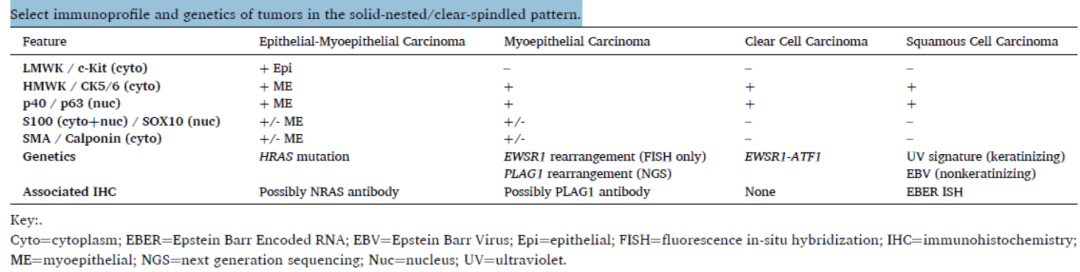
(Click to view larger image)
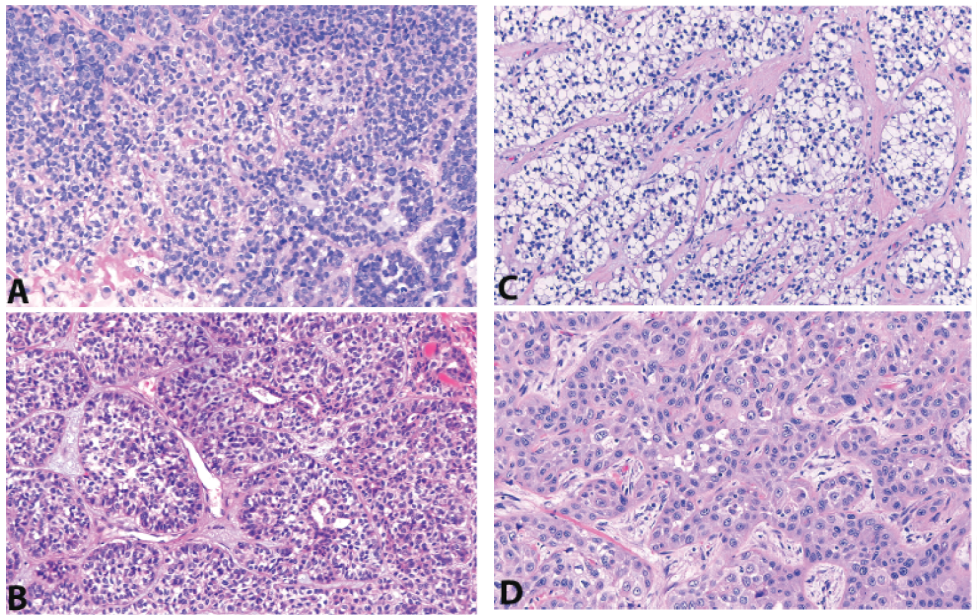
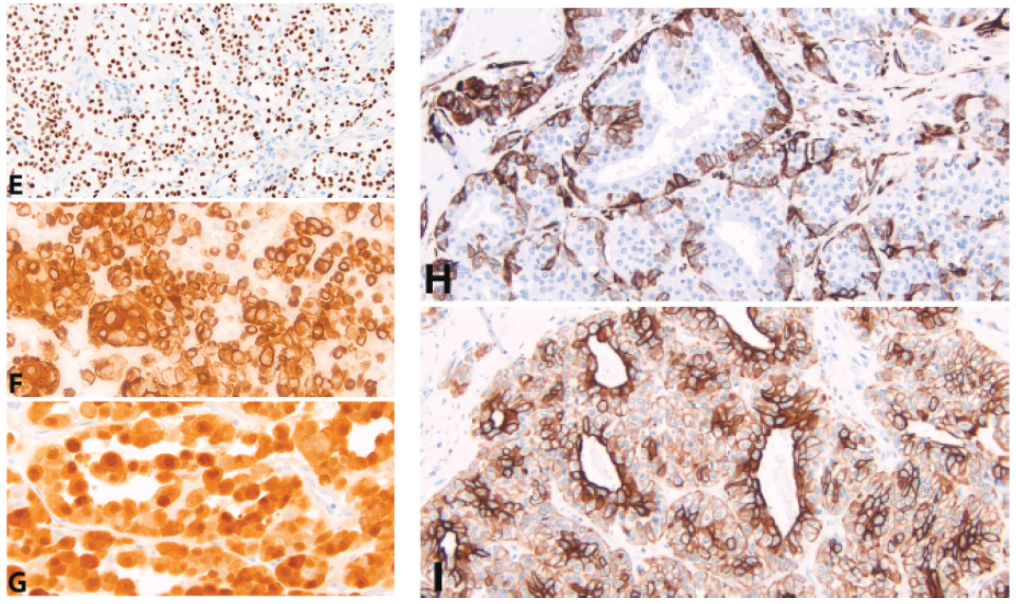
The immunophenotypes of these four tumors in this group show significant overlap [Figure 1]. Basal markers p40 and p63, as well as high molecular weight keratin (HMWK), are strongly and diffusely positive in CCC and SqCC.Other markers of myoepithelial differentiation, such as SMA, calponin, S100, and GFAP, are negative in these two tumors.Basal markers p40/p63, HMWK, epithelial membrane antigen (EMA), and myoepithelial markers can be variably positive in the myoepithelial population of MyoCA and EMC.Due to the high variability in the expression profile of tumor myoepithelial cells, a panel including all potential markers can be used to confirm myoepithelial differentiation:The presence of keratins (pankeratin, HMWK) or EMA plus basal or myoepithelial-specific markers (p40, p63, SMA, calponin, S100, GFAP) is confirmatory.Low molecular weight cytokeratin and c-Kit/CD117 are expressedin the epithelial cells of EMC, and should not show significant expression in MyoCA, CCC, or SqCC.Figure 1: Solid-Nested tumors. Many myoepithelial carcinomas (A) and epithelial-myoepithelial carcinomas (B) express p40/p63 (E) and high molecular weight keratin (in this case, CK5/6 (F)) as evidence of myoepithelial differentiation. Clear cell carcinoma (C) and squamous cell carcinoma (D) should both be positive. The myoepithelial cells of myoepithelial carcinoma and epithelial-myoepithelial carcinoma may also co-express S100 (G, myoepithelial carcinoma) and SMA (H, epithelial-myoepithelial carcinoma). Only epithelial-myoepithelial carcinoma shows a biphasic distribution of low molecular weight keratin-positive epithelial cells (I); however, in this case, the myoepithelial cells also show weaker CAM5.2 expression.
In the context of myoepithelial differentiation, SOX10 requires some discussion. SOX10 has been shown to be expressed in most salivary gland tumors and may not be helpful for differential diagnosis. However, SOX10 is negative in clear cell carcinoma, squamous cell carcinoma, salivary duct carcinoma, oncocytic tumors, and most mucoepidermoid carcinomas. Therefore, SOX10 may prove useful in specific contexts, particularly when negative.
Up to 40% of MyoCA with clear cytoplasmic changes and 80% of CCC have FISH rearrangements of EWSR1.
However, these MyoCA do not exhibit EWSR1 fusion transcripts in next-generation sequencing;instead, many have oncogenic PLAG1 fusions, either de novo or carcinoma ex PA.These new findings suggest that in MyoCA, EWSR1 abnormalities may be passenger mutations.On the other hand, most true CCCs do contain EWSR1-ATF1 gene fusions, with CREM being a less common partner.There is no immunohistochemical correlate for EWSR1 rearrangements.Studies have shown that EMC harbors HRAS Q61R/Q61K mutations, more frequently in de novo cases rather than carcinoma ex PA, which retains PLAG1 abnormalities.The SP174 antibody used to detect NRAS Q61R mutations has been shown to cross-react with HRAS and KRAS Q61R mutations in melanoma and medullary thyroid carcinoma; however, this antibody has not been studied in salivary gland tumors.Squamous cell carcinomas of the parotid gland mostly originate from metastases of primary skin lesions in the head (scalp, auricle, face, etc.).In most cases, the skin origin is confirmed by ultraviolet signature mutations.Squamous cell carcinoma with lymphoepithelial morphology may be EBV-positive and may represent a primary salivary gland tumor.In such cases, EBER in situ hybridization may be helpful.However, metastasis from the nasopharynx should always be excluded.To be




