
The diagnosis of salivary gland tumors is a challenge for surgical pathologists due to their low incidence and overlapping histological features. In small biopsies, the most critical information to convey for clinical management is distinguishing benign/low-grade tumors from high-grade carcinomas. This article will discuss the differential diagnosis of salivary gland tumors based on four broad morphological patterns: basaloid/tubular/cribriform, (micro)cystic/secretory/mucinous, solid nested/clear-spindle, and oncocytic/oncocytoid. With the aid of immunohistochemistry, identifying the number of cell types (primarily epithelial vs. myoepithelial/basal cells) can further subclassify tumors within these morphological categories. Additional tumor-specific immunomarkers are useful in certain cases. Underlying tumor-specific genetic abnormalities may be valuable; however, immunohistochemical correlations are only available for some. In the correct morphological context, when used appropriately and with an understanding of their limitations, immunohistochemical stains can help differentiate morphologically similar tumors.
Keywords: Salivary gland tumors, pattern-based, immunohistochemistry, genetics
Salivary gland tumors are rare, with up to 85% being benign, while salivary gland-type carcinomas account for only 6% of all head and neck tumors. The most common benign tumor is pleomorphic adenoma, and the most common malignant tumor is mucoepidermoid carcinoma, followed by adenoid cystic carcinoma. Many tumors, both benign and malignant, have significant morphological overlap, making definitive diagnosis challenging even in resection specimens. Identifying the unique histological features of each entity may be necessary to establish a diagnosis. This article will discuss the differential diagnosis of salivary gland tumors based on four major categories of morphological patterns (Figure 1). Within the context of these groupings, the selection of immunohistochemical stains may be approached differently. In some cases, targeted staining reflects the underlying genetic abnormalities diagnostic of specific tumors. The purpose of this review is to provide a pattern-based diagnostic approach to salivary gland tumors, using selective immunohistochemistry and genetics, when applicable, as ancillary techniques for differential diagnosis.
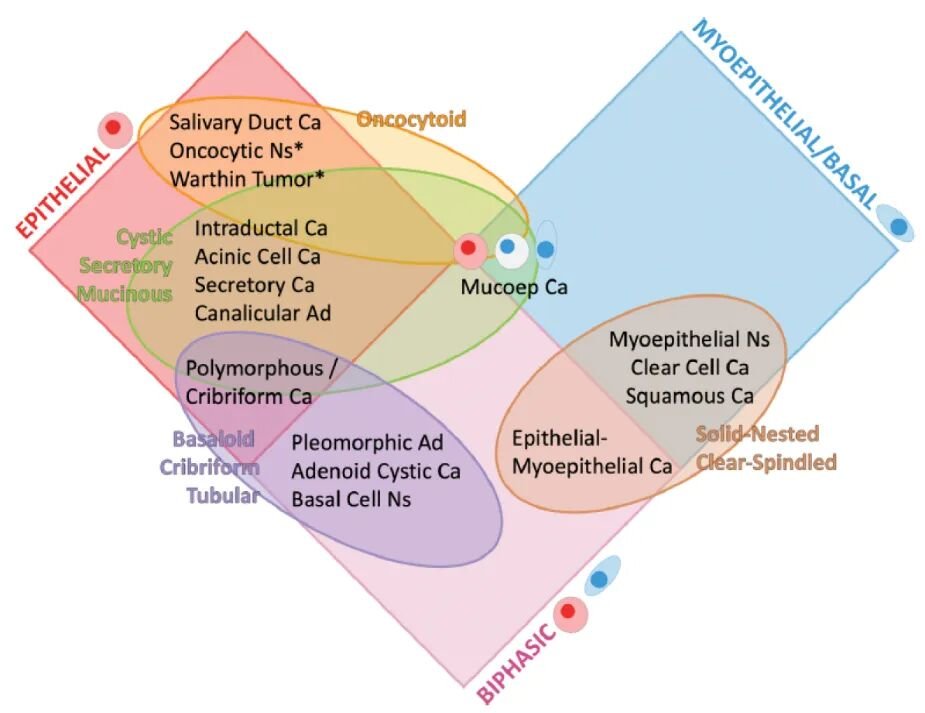
Figure 1: Overview of salivary gland tumors. Red diamonds include tumors with epithelial phenotype. Oncocytic tumors and Warthin tumor (*) are morphologically epithelial and monomorphic, demonstrated to have an outer layer of p63+. Blue diamonds include tumors with myoepithelial/basal phenotype. Magenta diamonds include biphasic tumors (epithelial and myoepithelial/basal cells). Orange ovals include tumors with oncocytic morphology. Green ovals include cystic/secretory/mucinous tumors. Purple ovals include tumors with basaloid/tubular/cribriform morphology. Brown ovals include solid-nested/clear-spindle category tumors. Key: Ca = carcinoma; ad = adenoma; Ns = neoplasm.
Biopsies of major salivary glands are typically performed using fine-needle aspiration (FNA) or core needle biopsy, while biopsies of minor salivary glands within the oral cavityare usually performed via incisional biopsy or curettage biopsy. Core, incisional, or curettage biopsies are often small, may have distorted architecture, and may lack interfaces with normal structures. Preoperative biopsies should aim to determine two items. First, whether the lesion is more likely a primary salivary gland tumor or a metastasis. There are lymph nodes within and around major salivary glands, and local metastasis should be considered in the differential diagnosis. Second, whether the lesion is high-grade or low-grade. Both benign tumors and low-grade carcinomas typically require local surgical excision with negative margins. High-grade carcinomas usually require additional surgical planning and neck dissection. High-grade features in many salivary gland tumors include marked nuclear atypia, necrosis, increased or atypical mitoses, and perineural or lymphovascular invasion. Diagnoses from core, incisional, or curettage biopsies are often descriptive, accompanied by grading. Definitive subclassification is best performed on surgical resection specimens, where the entire tumor can be examined and supplemented with ancillary tests as needed.
Familiarity with the microanatomy of the salivary gland helps correlate salivary gland morphology with immunohistochemistry. Normal ducts and acini have luminal epithelial cells and abluminal cells, which are contractile myoepithelial cells or non-contractile basal cells. The epithelial component of each acinus consists of serous cells and/or mucous cells. Contractile myoepithelial cells surround individual acini. There is a morphological transition from the outer myoepithelial cell layer to the outer basal cell layer: intercalated ducts (small ducts between acini) have a simple cuboidal epithelial inner layer and an outer myoepithelial layer. Larger striated ducts are lined by simple columnar cells, with a transition from peripheral myoepithelial cells to basal cells. Large interlobular/excretory ducts are lined by pseudostratified columnar cells surrounded by basal cells. The inner luminal epithelial cells tend to express low molecular weight cytokeratins. The outer abluminal myoepithelial and basal cells express high molecular weight cytokeratins and basal cell markers (p40/p63). Specific myoepithelial markers (SMA, calponin, S100, SOX10) are expressed in cells with myoepithelial differentiation (but not basal differentiation).
When applied in the context of morphological patterns, identifying the number and types of cells can aid in differential diagnosis. Additional stains, either specific to tumors or reflecting underlying molecular genetics, can further assist in classification. This review is not an exhaustive discussion of the morphology and immunophenotype of all salivary gland tumors but will focus on selected immunostaining panels that help analyze common tumors within the four main morphological pattern groups: basaloid/cribriform/tubular, (micro)cystic/secretory/mucinous, solid-nested/clear-spindle, and oncocytic/oncocytoid. In some cases, confirming underlying genetic abnormalities via polymerase chain reaction (PCR), fluorescence in situ hybridization (FISH), or next-generation sequencing (NGS) may help support a definitive diagnosis.
Basaloid/Cribriform/Tubular
This category of tumors (Table 1) appears blue, basaloid, and often monotonous at low magnification. Typically, these tumorshave high cellular density and a high nuclear-to-cytoplasmic ratio.Their architecture is diverse, including cribriform, tubular, and solid growth patterns, occasionally with a mucinous stromal background.This category includes adenoid cystic carcinoma (AdCC), basal cell neoplasms (BCN), polymorphous adenocarcinoma (PAC), and cellular pleomorphic adenoma (PA).AdCC is biphasic, tending to have a distinctive cribriform architecture with associated blue-gray mucoid matrix material.BCN (adenoma and adenocarcinoma) are also biphasic, presenting as large nests (so-called membranous or jigsaw) to small tubular-trabecular patterns with peripheral palisading, dense eosinophilic basement membrane-like material arranged in globules or bands.Basal cell adenoma and adenocarcinoma are included together in the BCN subclass because the distinction between them, based on invasiveness, is made upon complete histological examination.PAC has a single cell population and typically features ground-glass nuclei/wrinkling, reminiscent of papillary thyroid carcinoma.The architecture ishighly variable and can include cartwheel, single-file, or glomeruloid patterns.The cribriform variant is considered together with the classic PAC subtype due to their similar immunophenotype.Finally, cellular PA is biphasic and must contain bilayered ducts.Diagnosis is challenging if sheets or cribriform structures predominate and typical chondroid stroma is sparse or absent.As confounding factors, PA can also exhibit mucinous, sebaceous, squamous, oncocytic, and lipomatous metaplasia.Pure myoepithelioma also deserves mention in this category; however, there is interobserver variability between duct-poor/rich myoepithelial PA and myoepithelioma, depending on the pathologist’s threshold for the allowable number of ducts.Finally, although epithelial-myoepithelial carcinoma may also enter the differential as a biphasic tumor, it is typically dominated by myoepithelial cells and will therefore be discussed in the solid-nested/clear-spindle chapter.
Table 1. Immunophenotype and genetics of selected basaloid/cribriform/tubular tumors

(Click to view larger image)
AdCC, BCN, and PA contain a p40/p63-positive basal/myoepithelial cell population (Figures 2 and 3). In AdCCand PA, this cell population is present in the abluminal cell layer of tubules, with the luminal epithelial layer expressing CD117/c-Kit.In cribriform areas, epithelial cells are sandwiched between myoepithelial cells, which are present both at the outer edges of cribriform nests and lining the cribriform spaces containing blue mucoid material.It is this map-like expression pattern that is most helpful.Solid areas of AdCC may diffusely express CD117.In BCN, p40/p63 tends to be expressed in the palisaded peripheral basal cell layer of nests.PAC is the only tumor in this group with a single cell population that does not express p40.The absence of p40 expression is most helpful in distinguishing PAC, as interestingly, p63 canbe expressed in a patchy distribution.CD117/c-Kit is often positive in basal cell tumors, especially in central epithelial cells, and shows a non-specific staining pattern in PAC.Although CD117 is generally considered a marker for AdCC, it should be interpreted with caution as it is also expressed in other salivary gland tumors, as previously mentioned.
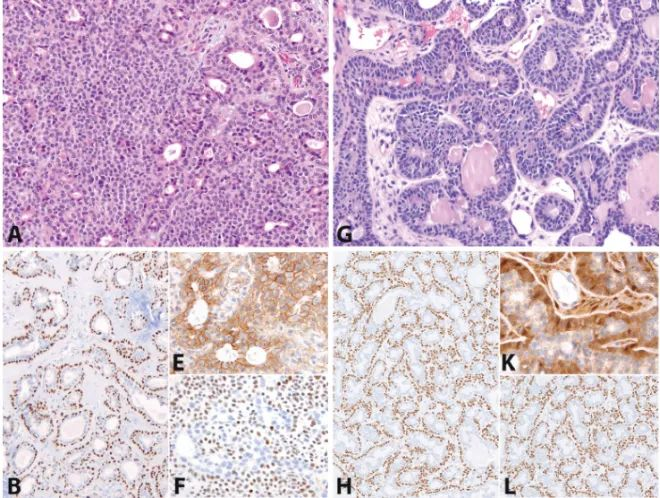
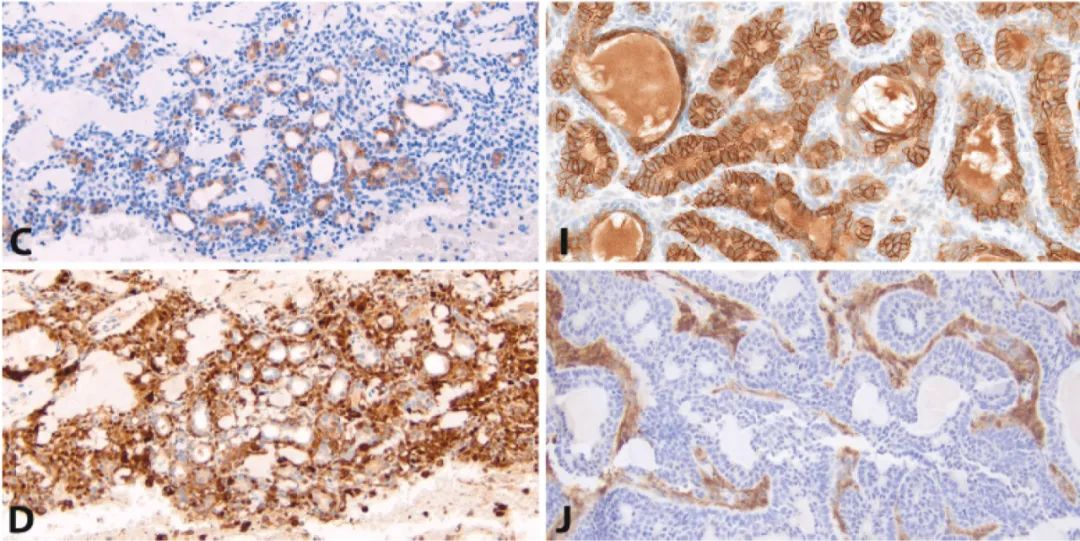
Figure 2: Pleomorphic adenoma vs. basal cell neoplasm. Cellular pleomorphic adenoma (A) and basal cell neoplasm (G) both show p40/p63-positive peripheral cells (B and H) and CD117-positive central epithelial cells (C and I). S100 often highlights myoepithelial cells around ducts or in sheets of pleomorphic adenoma (D). S100 can show non-specific staining of epithelial-like nests in tubular-trabecular basal cell neoplasms but strongly highlights the inter-nest spindle cell stroma (J). Many CTNNB1-mutated basal cell adenomas express β-catenin in the nuclei/cytoplasm of peripheral cell layers and spindle stroma (K). Other CTNNB1 wild-type tumors show only membranous expression (E). LEF1 can be expressed in both basal cell (L) and non-basal cell (F) tumors.
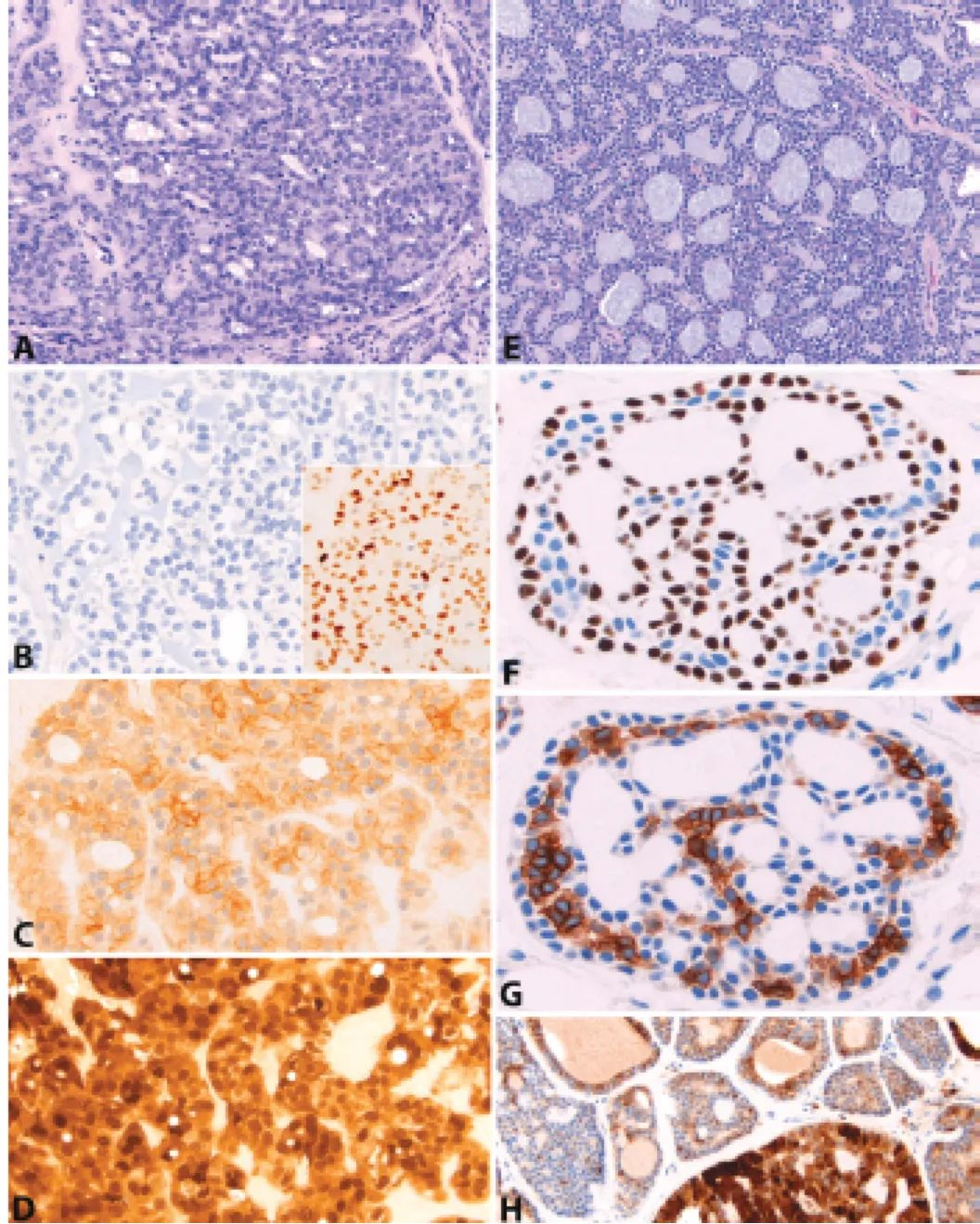
Figure 3: Polymorphous/cribriform adenocarcinoma vs. adenoid cystic carcinoma. Polymorphous adenocarcinoma (A) and its cribriform variant are both negative for p40 (B), confirming the lack of a true basal cell population. However, p63 can show patchy expression (B, inset). CD117 can also show weak patchy expression (C). S100 is typically strongly and diffusely positive (D). In contrast, adenoid cystic carcinoma (E) shows a map-like distribution of p40 and p63-positive cells at the peripheral basement membrane and central stromal areas (F), with CD117-positive epithelial cells sandwiched in between (G). S100 can show variable non-specific expression (H).
Another immunostain crucial for PAC is S100, which shows strong and diffuse expression [Figure 3]. AdCC can exhibit non-specific patchy S100 expression without a specific map-like distribution. In PA, S100 is typically confined to myoepithelial cells and not expressed in luminal epithelial cells. However, hyaline or plasmacytoid myoepithelial cells in PA can show strong S100 expression, posing a diagnostic dilemma if overrepresented. Although plasmacytoid cells can occur in PAC, they are more common in PA. In BCN, S100 is strongly expressed in the spindle cell stroma and can be patchy in epithelial-like nests.
When morphological features and immunoprofiles are ambiguous, genetics may aid in diagnostic confirmation. AdCC typically harbors NFIB rearrangements, most commonly partnered with MYB, followed by MYBL1. MYB protein overexpression, which can be confirmed by immunostaining, is mainly in the nuclei of myoepithelial cells. However, nuclear expression is rare in other basaloid tumors (BCN, PAC, PA), so genetics may be more specific. Less commonly, NOTCH mutations are detected in AdCC, most frequently NOTCH1. Diffuse nuclear NOTCH1 expression correlates with NOTCH1 mutation status and predicts worse clinical outcomes. However, mottled or so-called ‘subset’ expression can occur in NOTCH1 wild-type AdCC and non-AdCC tumors (including BCN, PAC, and PA), which may limit its utility. CLYD1 gene mutations may exist in basal cell tumors; however, this mutation has not been shown to correlate with immunostaining. Basal cell adenomas more commonly have CTNNB1 mutations; subsequently, BCN may show nuclear expression of β-catenin in basal cell populations and stromal cells. Non-BCN generally show only membranous expression. Basal cell tumors can also show nuclear positivity for LEF-1 (a β-catenin co-activator), but this marker is not specific and can also be positive in PA, AdCC, epithelial-myoepithelial carcinoma, and clear cell carcinoma. [Figure 2] Polymorphous adenocarcinoma harbors PRKD1 point mutations. The cribriform variant often contains PRKD1, 2, or 3 gene rearrangements. However, there is no immunohistochemical correlation for PRKD abnormalities. Pleomorphic adenomas often have alterations in PLAG1 and HMGA2; PLAG1 and HMGA2 immunostaining indicates these genetic changes in the tumor. However, these markers may be positive in PAC, BCN, myoepithelial carcinoma, and epithelial-myoepithelial carcinoma but may not be sensitive or specific enough.







