Application and Pitfalls of Immunohistochemical Antibodies in Diagnosing Urogenital Diseases (Part 2)
Introduction:
The application of immunohistochemical antibodies in the genitourinary system is becoming increasingly common. When biopsy tissue is very limited, selecting the optimal IHC markers becomes even more important. In the previous article, we shared the application and pitfalls of immunohistochemistry in diagnosing prostate diseases. This article will focus on the application and pitfalls of immunohistochemistry in diagnosing bladder diseases.
Bladder
|
Table1 Bladder Markers |
|||
|
Marker |
Benign Reactive Urothelium |
Urothelial Carcinoma |
Bladder Adenocarcinoma |
|
CK20 |
Umbrella Cells |
+ |
+ |
|
P63 |
+ |
+ |
- |
|
GATA3 |
+ |
+ |
- |
|
CD44 |
Full-thickness |
- |
+/- |
|
p53 |
Basal Cells |
Strongly Positive |
+/- |
|
β-Catenin |
- Non-nuclearstaining |
- Non-nuclear Staining |
- Non-nuclear Staining |
|
CDX2 |
- |
- |
+ |
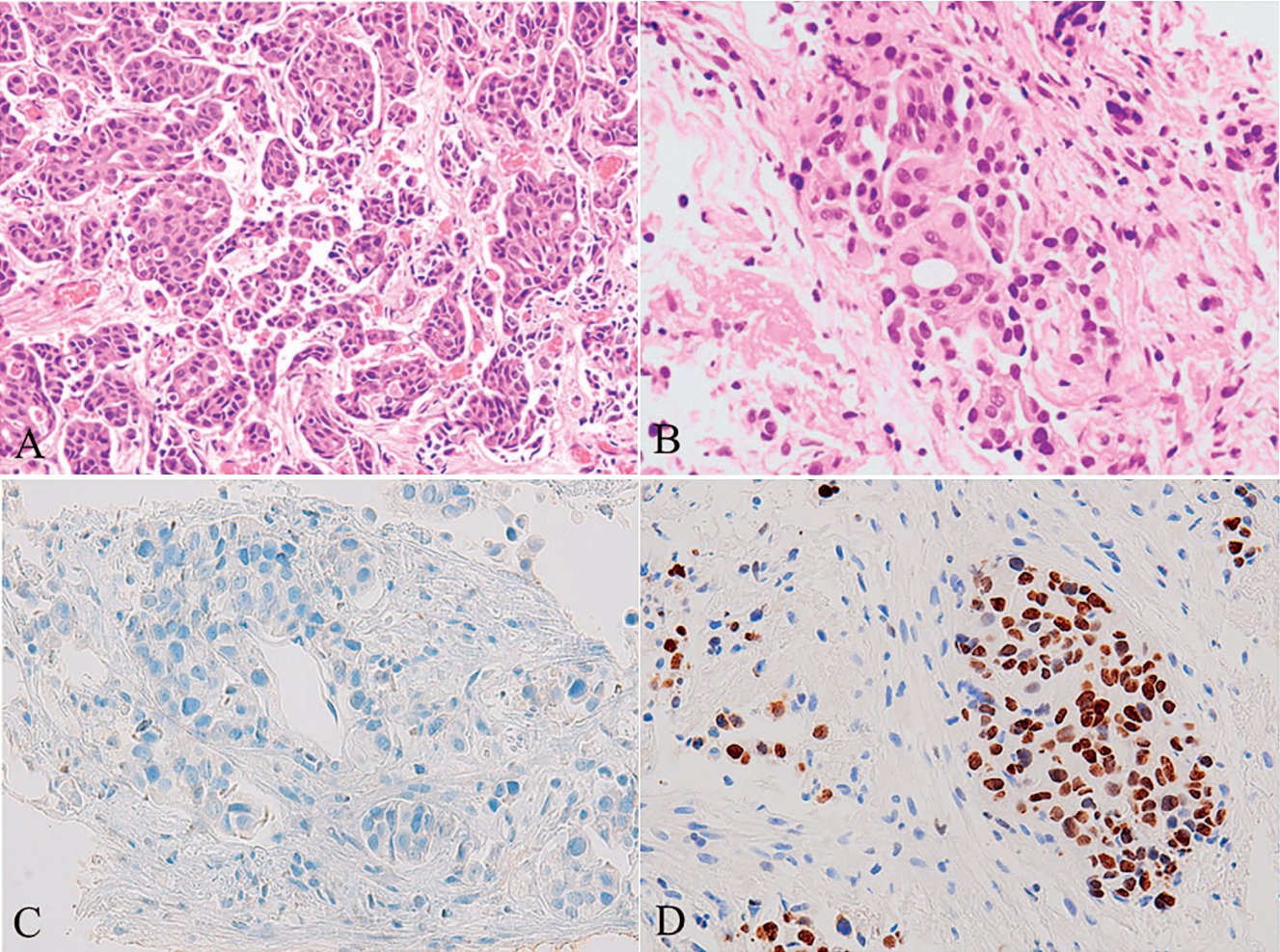
Figure 1A. Bladder Micropapillary Urothelial Carcinoma
Figure 1B. Bladder Micropapillary Urothelial Carcinoma. Clusters of tumor cells are embedded in ‘lacunae,’ and these tumor cells are relatively bland in morphology.
Figure 1C. Bladder Micropapillary Urothelial Carcinoma. p63 staining shows loss of basal cells.
Figure 1D. Bladder Micropapillary Urothelial Carcinoma. Positive expression of GATA3.
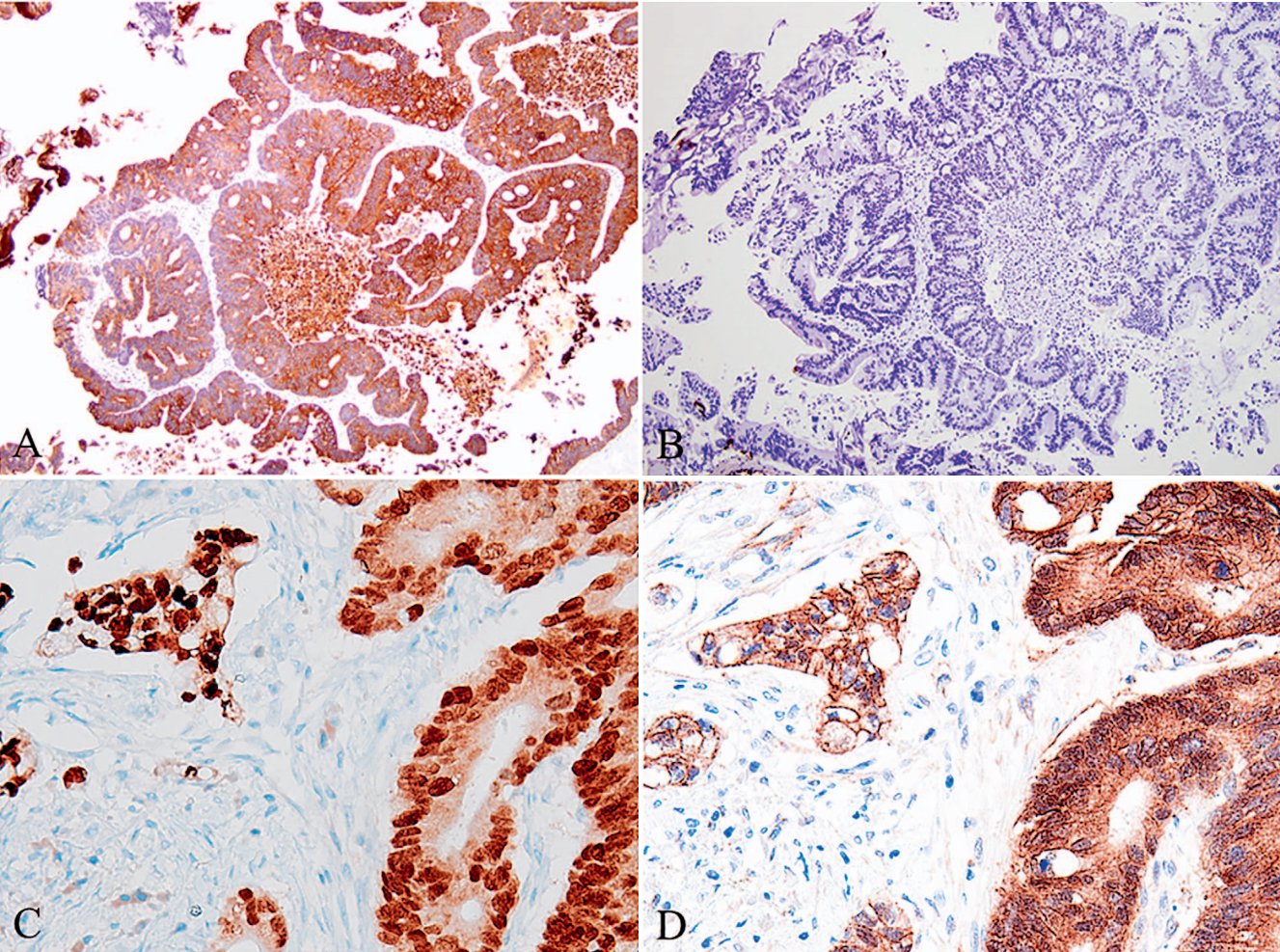
Figure 2A. Primary Bladder Adenocarcinoma. Similar to the immunophenotype of colorectal cancer, primary bladder adenocarcinoma (intestinal type) is CK20 positive;
Figure 2B. Primary Bladder Adenocarcinoma. Similar to the immunophenotype of colorectal cancer, primary bladder adenocarcinoma (intestinal type) is CK7 negative;
Figure 2C. Primary Bladder Adenocarcinoma. Similar to the immunophenotype of colorectal cancer, primary bladder adenocarcinoma (intestinal type) is CDX2 positive;
Figure 2D. Primary Bladder Adenocarcinoma. Primary bladder adenocarcinoma shows cytoplasmic staining for β-catenin, but lacks the nuclear β-catenin staining commonly seen in colorectal cancer.
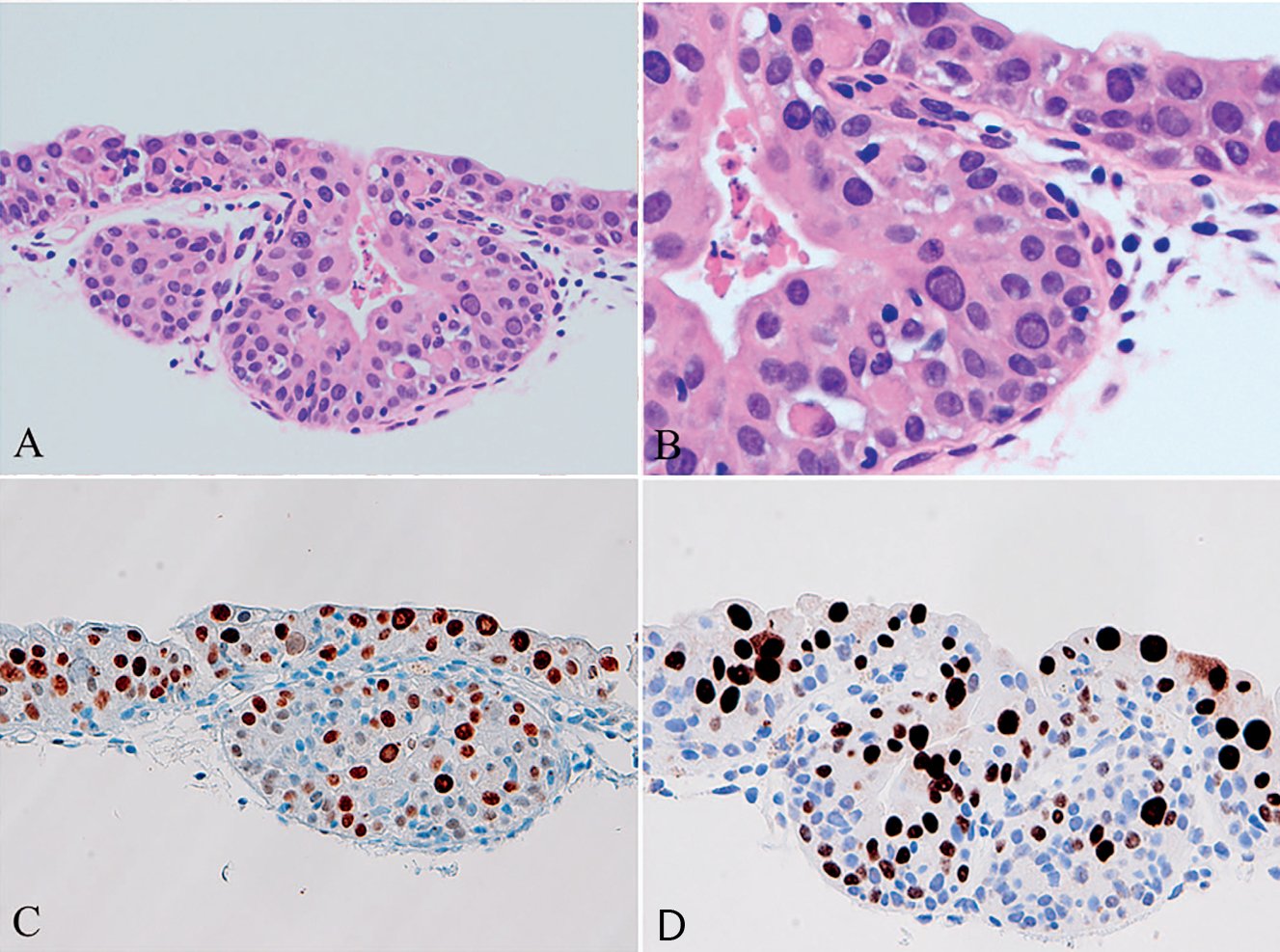
Figure 3A. Cytological Atypia due to BK Virus. Urothelial cells infected with BK virus;
Figure 3B. Cytological Atypia due to BK Virus. Marked cytological atypia, mimicking urothelial carcinoma in situ;
Figure 3C. Cytological Atypia due to BK Virus. Very high proliferative activity of Ki-67;
Figure 3D. Cytological Atypia due to BK Virus. Positive staining with SV40 antibody cross-reactive with BK virus.
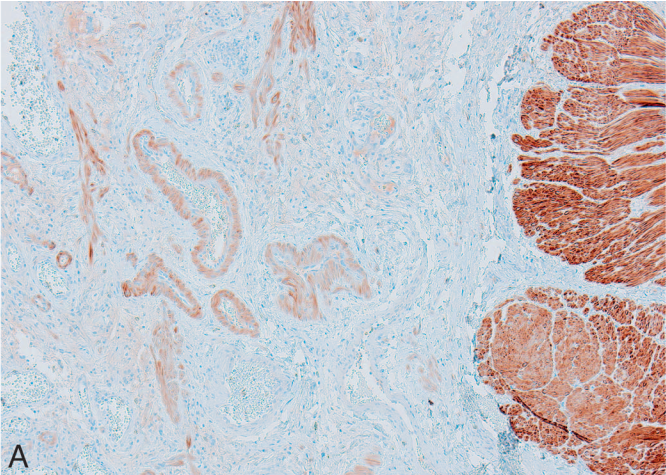
Figure 4A. Application of Smoothelin in Bladder Cancer. Smoothelin is strongly positive in the muscularis propria (right side of Figure 8A, Smoothelin stain) and weakly positive in the muscularis mucosae and smooth muscle bundles of background vessels (left side of Figure 8A, Smoothelin stain).
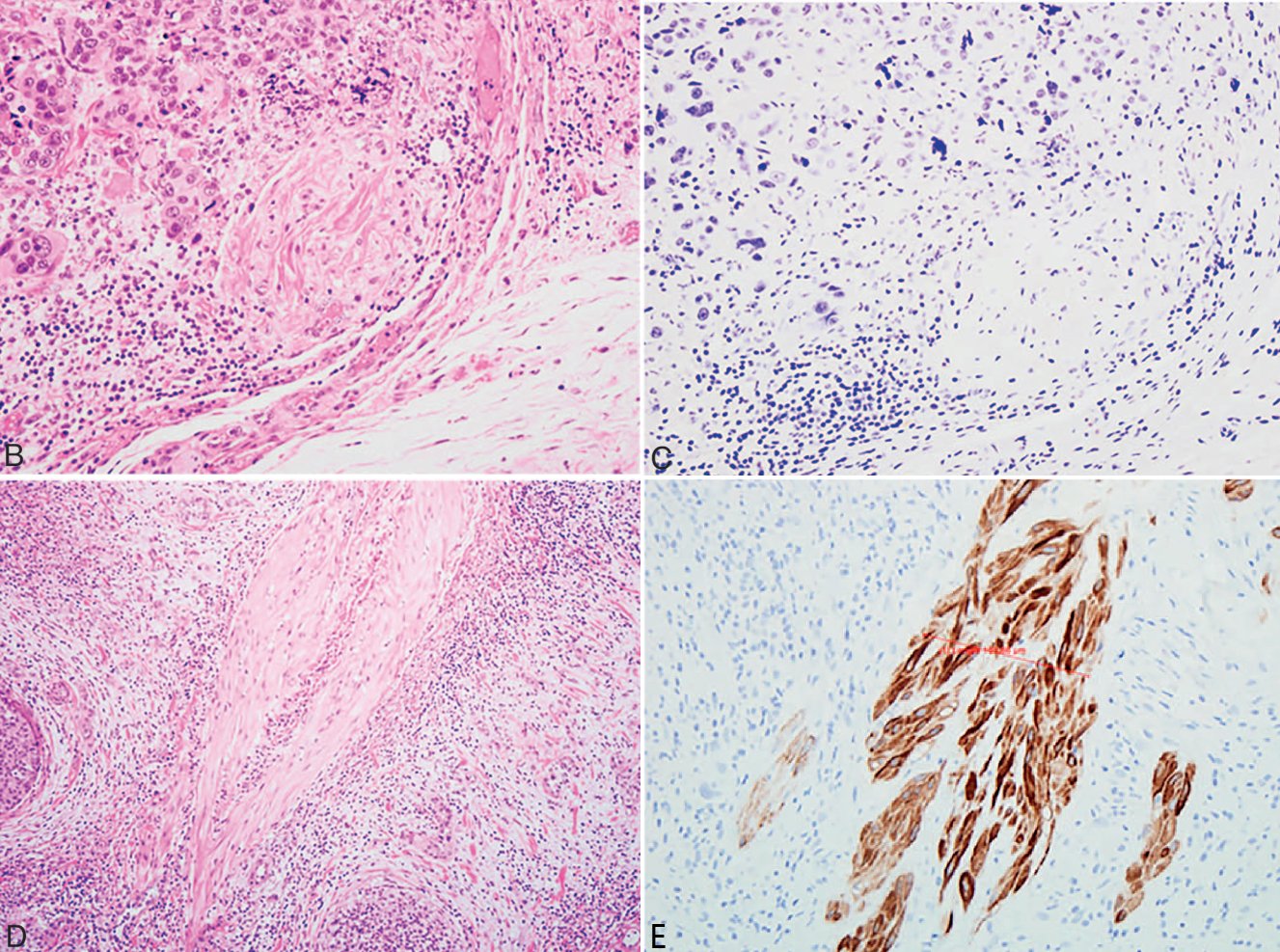
Figure 4B. Application of Smoothelin in Bladder Cancer. Urothelial carcinoma cells invading smooth muscle bundles;
Figure 4C. Application of Smoothelin in Bladder Cancer. Same field as Figure 8B, showing faint smoothelin staining, suggesting involvement of the muscularis mucosae;
Figure 4D. Application of Smoothelin in Bladder Cancer. Another case of urothelial carcinoma invading smooth muscle bundles;
Figure 4E. Application of Smoothelin in Bladder Cancer. Same field as Figure 8D, showing strong smoothelin positivity, suggesting involvement of the muscularis propria.
Maxin Related Antibodies
|
Antibody Name |
Product Code |
Clone Number |
Positive Localization |
|
CK20* |
MAB-0834 |
MX059 |
Cytoplasmic |
|
CDX2* |
MAB-0713 |
MX024 |
Nuclear |
|
p53* |
MAB-0674 |
MX008 |
Nuclear |
|
p63* |
MAB-0694 |
MX013 |
Nuclear |
|
GATA3 |
MAB-0695 |
L50-823 |
Nuclear |
|
β-Catenin* |
MAB-0754 |
MX043 |
Cytoplasmic/Membranous |
*Indicates Maxin clone product
Jenny Ross, Guangyuan Li, Ximing J. Yang. Application and Pitfalls of Immunohistochemistry in Diagnosis of Challenging Genitourinary Cases. Arch Pathol Lab Med. Vol 144, March 2020:290-304
