High-grade renal cell tumors, or when they appear as metastases, also present diagnostic pitfalls. Table 1 summarizes the commonly used immunohistochemical markers for renal tumors. The most well-known antibodies include the MiTF (microphthalmia-associated transcription factor) translocation family with TFE3 (transcription factor binding to IGHM enhancer 3) or TFEB (transcription factor EB) translocations.

In general, clear cell RCC should bePax–8positive, CA-IX (carbonic anhydrase IX) positive, and CK7 negative. AMACR can be positive in a small subset of clear cell RCC, but its expression pattern is usually weak and focal; papillary RCC is positive for Pax-8, AMACR, and CK7, and negative for CA-IX (Table 1). High-grade type 2 papillary RCC often shows focal CK7 staining. When large papillary RCCs undergo extensive necrosis due to ischemia, they may express CA-IX due to normal biological cell signaling, and this should not be interpreted as clear cell RCC. Conversely, clear cell RCC may show focal or patchy expression of CK7 or AMACR. Accurate diagnosis requires integrating the overall immunohistochemical expression pattern and depends on extensive sampling. CD10 has limited utility in RCC diagnosis because it is positive in most RCC subtypes; similarly, due to its low specificity, CD10 should be used sparingly in metastatic tumors.
Renal angiomyolipomas can express HMB-45, Melan-A, MiTF, tyrosinase, and one or more actins, making diagnosis relatively straightforward in typical cases. However, diagnosing epithelioid angiomyolipoma can be challenging. Epithelioid angiomyolipoma has two main architectural patterns: ① Carcinoma-like growth pattern, characterized by large cells arranged in broad alveolar structures, growing in nests or sheets with rich vascular septa; ② Diffuse growth pattern, consisting of sheets of epithelioid to plump spindle cells, which may contain multinucleated giant cells and melanin pigmentation, occasionally with necrosis and microabscesses. Five clinicopathological features are associated with aggressive behavior in epithelioid angiomyolipoma: (1) association with tuberous sclerosis; (2) tumor size larger than 7 cm; (3) carcinoma-like growth; (4) involvement of perirenal soft tissue and/or kidney; (5) necrosis (Figure 1). Therefore, it is important to provide these key diagnostic findings to clinicians for selecting the most successful treatment plan for the patient. Cathepsin K is a cysteine protease that shows strong cytoplasmic positivity in renal angiomyolipomas, including epithelioid subtypes, oncocytoma-like, and epithelioid cystic subtypes, which may be negative for melanocytic or myogenic markers. Cathepsin K is also well-expressed in perivascular epithelioid cell tumors (PEComas) at other body sites. Differential diagnosis includes translocation-associated RCC because it can express Cathepsin K.

Figure 1A. Epithelioid angiomyolipoma. The tumor measures 15 cm in greatest dimension, replacing the entire kidney;
Figure 1B. Epithelioid angiomyolipoma. The tumor cells show diffuse growth with necrosis;
Figure 1C. Epithelioid angiomyolipoma. Higher magnification shows large epithelioid tumor cells with clear to eosinophilic cytoplasm, resembling renal cell carcinoma;
Figure 1D. Epithelioid angiomyolipoma. The tumor cells are positive for HMB-45 but negative for epithelial and renal cell markers.
Renal medullary carcinoma is a rare aggressive tumor, and patients almost always have sickle cell trait. SMARCB1 (also known as INI1) is a core subunit of the SWI/SNF chromatin remodeling complex, and loss of this protein expression is a key diagnostic feature of renal medullary carcinoma. Renal medullary carcinoma is typically positive forPax–8, CK7, and OCT4, and may express CK20 and Vimentin. However, besides renal medullary carcinoma, other renal tumors such as pediatric rhabdoid tumor and epithelioid sarcoma, and even collecting duct carcinoma, may also show loss of SMARCB1.
Compared to classic RCC, translocation-associated RCC has multiple relatively unique morphological patterns within the same tumor, including papillary architecture, clear cytoplasm, high nuclear grade, psammoma bodies, and sometimes rosette formation. Unlike clear cell RCC or papillary RCC, translocation-associated RCC is typically positive forPax–8but usually negative for CA-IX and/or AMACR. Additionally, although clear cell RCC may rarely express weak Melan-A, a small subset of translocation-associated carcinomas can be positive for Melan-A or HMB-45, which is uncommon in other types of RCC. In such cases, TFE3 immunohistochemistry should be added (Figure 3, B to E). However, the TFE3 antibody is technically challenging to work with, having significant background staining and poor specificity.

Figure 2A. TFE3 translocation-associated renal cell carcinoma. Grossly, the tumor is large with areas of hemorrhage and necrosis, almost completely replacing the entire kidney;
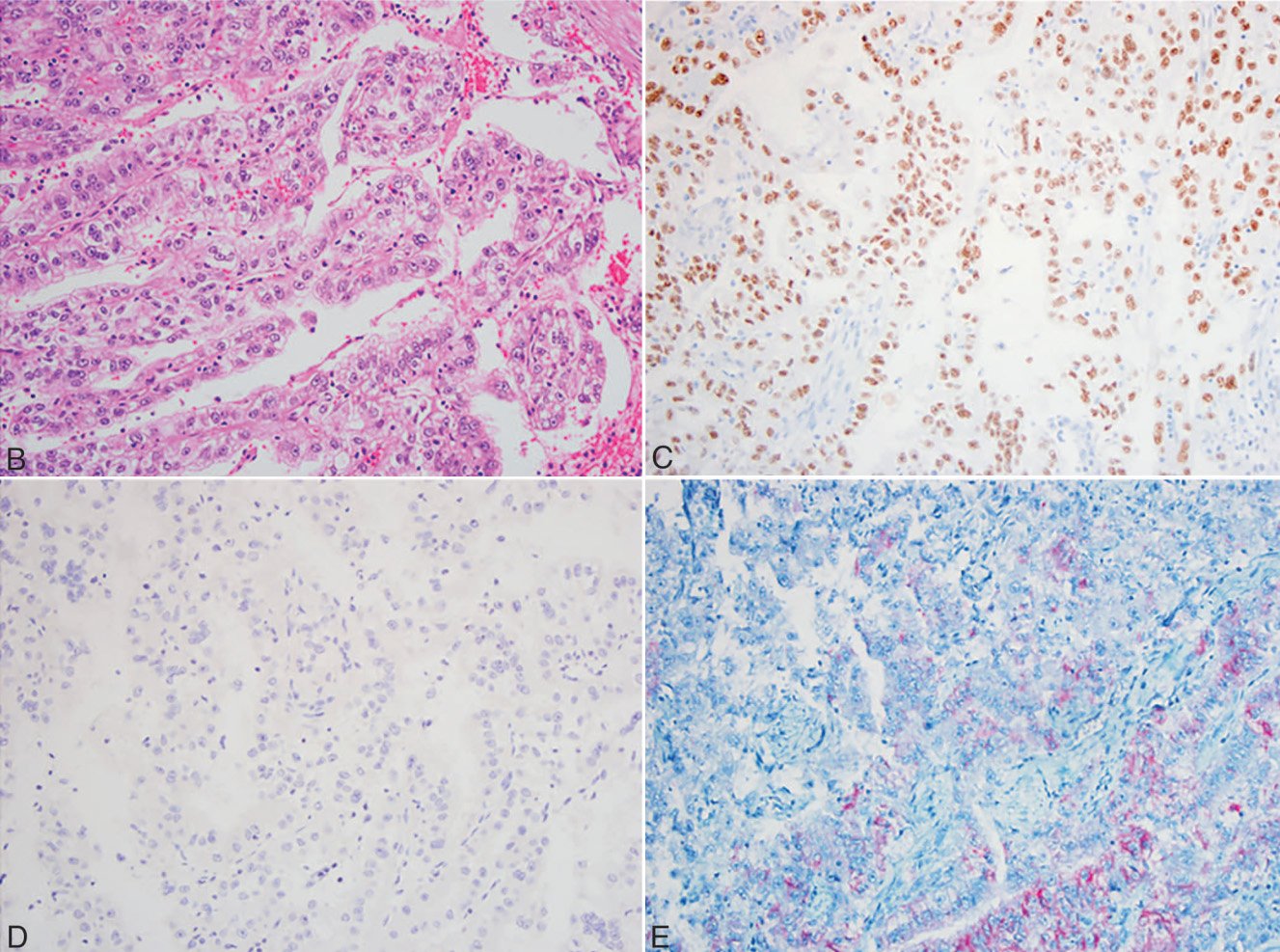
Figure 2B. TFE3 translocation-associated renal cell carcinoma. Microscopically, the tumor is composed of high-grade tumor cells with clear cytoplasm, forming glandular and papillary structures;
Figure 2C. TFE3 translocation-associated renal cell carcinoma. The tumor cells are positive for TFE3;
Figure 2D. TFE3 translocation-associated renal cell carcinoma. The clear cell RCC marker CA-IX is negative;
Figure 2E. TFE3 translocation-associated renal cell carcinoma. The tumor cells also show focal positive expression for Melan-A.
When a tumor is located in the central part of the kidney, differential diagnosis with UC is necessary. UC can show extensive clear cell change. UsingPax–8can demonstrate renal origin, but some upper tract UC may express weak to moderatePax–8, andPax–8may also be expressed in sarcomatoid subtypes of renal and urothelial tumors. Differential diagnosis is particularly difficult when the specimen is limited. In such cases, an immunohistochemical panel includingPax–8, p63, GATA3, and S100P can help differentiate them.
The newly added eosinophilic solid and cystic renal cell carcinoma (ESC RCC) is a recently recognized distinct type of indolent renal tumor occurring in female patients with or without tuberous sclerosis. It is characterized by tumor cells with prominent eosinophilic cytoplasm and granular cytoplasmic stippling, with round to oval nuclei (Figure 3, A and B). The tumor shows a mixture of solid and cystic areas, with positive expression forPax–8and CK20, focal positivity for AMACR (Figure 3, C and D), and negativity for CD117 and CK7.

Figure 3A. Eosinophilic solid and cystic renal cell carcinoma. The tumor consists of cystic areas (Figure 11A) and solid areas (Figure 11B);
Figure 3B. Eosinophilic solid and cystic renal cell carcinoma. The tumor cells have prominent eosinophilic cytoplasm and granular cytoplasmic stippling (Figure 3B, arrow);
Figure 3C. Eosinophilic solid and cystic renal cell carcinoma. The tumor cells are positive for CK20;
Figure 3D. Eosinophilic solid and cystic renal cell carcinoma. The tumor cells show focal positivity for AMACR.
Most markers used to evaluate testicular germ cell tumors have some degree of overlapping expression ranges (Table 2). Seminomas and intratubular germ cell neoplasia are generally positive for CD117, and most germ cell tumors express OCT4. Embryonal carcinoma is positive for CD30 and OCT4. Both seminoma and embryonal carcinoma can express D2-40, but they show different staining patterns: seminoma exhibits diffuse membranous D2-40 staining, while embryonal carcinoma shows focal lacelike D2-40 staining (Figure 4). Yolk sac components express AFP and Glypican-3. Glypican-3 is also expressed in trophoblastic cells. Choriocarcinoma is positive for β-hCG (beta subunit of human chorionic gonadotropin; Figure 4, A and B). A small subset of seminomas may secrete β-hCG or contain syncytiotrophoblast cells secreting β-hCG, which should not be misdiagnosed as choriocarcinoma.
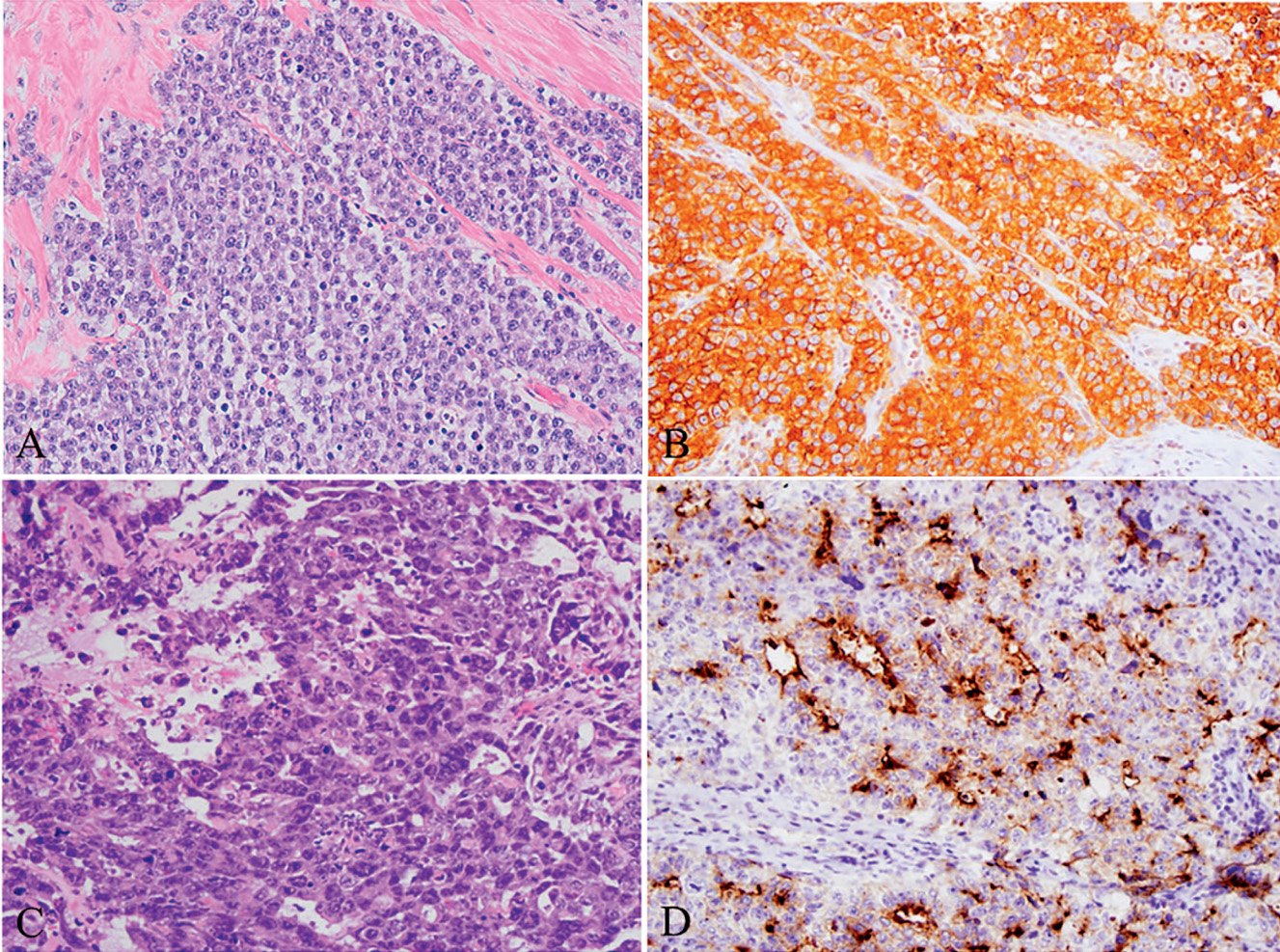
Figure 4A. Classic seminoma;
Figure 4B. Classic seminoma shows diffuse positivity for D2-40;
Figure 4C. Embryonal carcinoma;
Figure 4D. Embryonal carcinoma, showing lacelike D2-40 staining.
SALL4 is another marker for germ cell tumors (C and D in Figure 5), labeling seminoma, most embryonal carcinomas, yolk sac tumors, and teratomas; it is rarely expressed in non-germ cell tumors.
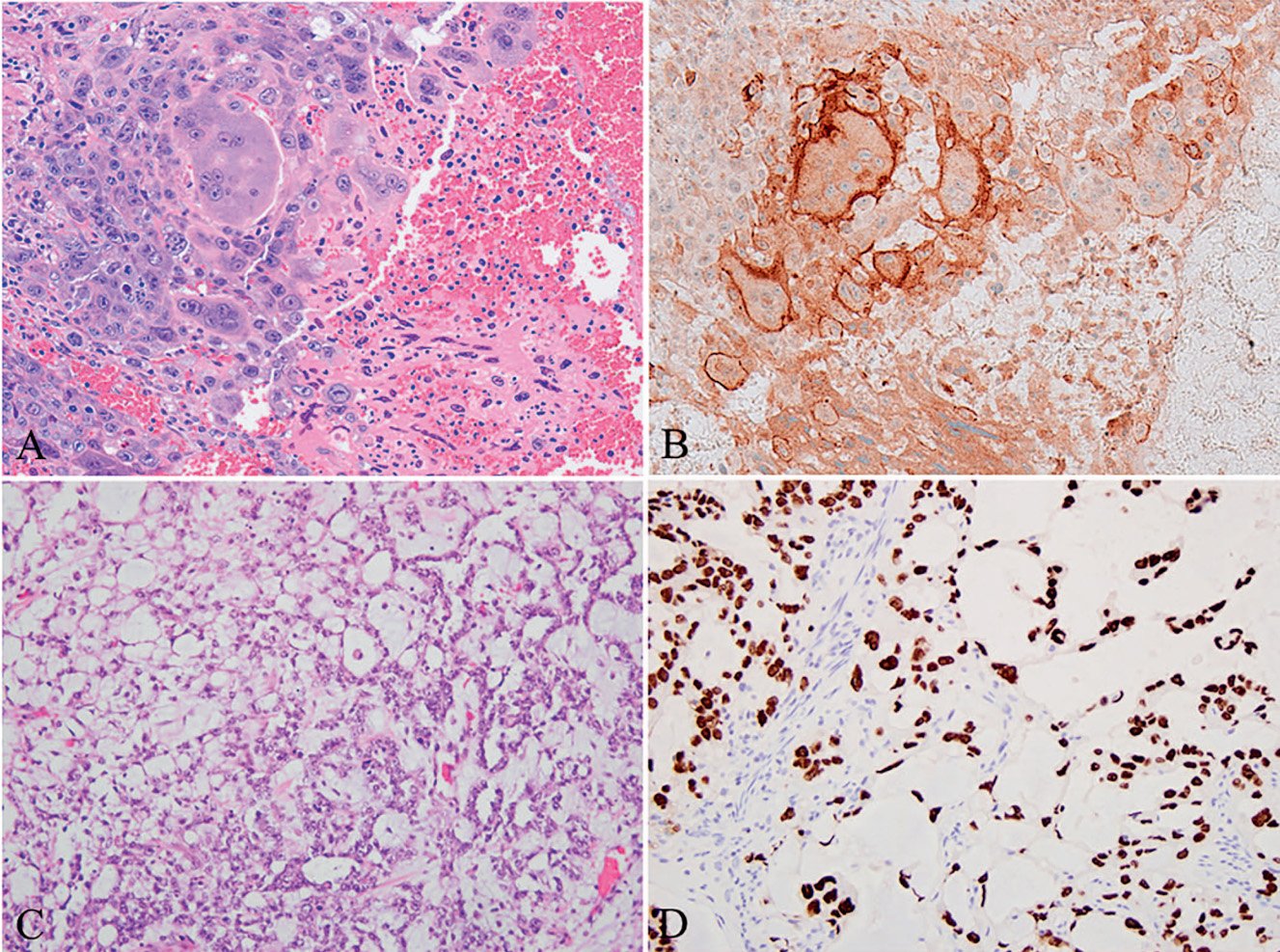
Figure 5A. Syncytiotrophoblast cells of testicular choriocarcinoma;
Figure 5B. Syncytiotrophoblast cells of testicular choriocarcinoma. Positive for Glypican-3;
Figure 5C. Yolk sac tumor;
Figure5D. Yolk sac tumorSALL4positive.
If a testicular tumor is found in elderly individuals, a panel of hematopoietic markers such as CD45, CD20, and CD3 should be added. For example, myeloid sarcoma can express CD117, and embryonal carcinoma expresses broad-spectrum cytokeratin, which may lead to misdiagnosis. Without other hematopoietic markers, hematopoietic tumors might be misdiagnosed as seminoma or embryonal carcinoma.
|
Antibody Name
|
Product Number
|
Clone Number
|
Positive Location
|
|
CA IX
|
RAB-0615
|
Polyclonal
|
Membrane
|
|
D2-40
|
MAB-0567
|
D2-40
|
Cytoplasm/Membrane
|
|
Glypican-3*
|
MAB-0667
|
MAXIM001
|
Cytoplasm
|
|
INI-1
|
MAB-0696
|
MRQ-27
|
Nucleus
|
|
Pax-8*
|
MAB-0837
|
MX062
|
Nucleus
|
|
TFE3
|
MAB-0663
|
MRQ-37
|
Nucleus
|








