Neurotrophic tyrosine receptor kinase (Neurotrophic Tyrosine Receptor Kinase, abbreviated as NTRK) gene fusion mutations have led to the development of drugs with significant clinical efficacy, both approved and under investigation. Many advanced patients have experienced remarkable recoveries after using NTRK inhibitors. So, what is the relationship between NTRK and its encoded protein: tropomyosin receptor kinase (Tropomyosin receptor tyrosine kinase, abbreviated as TRK) and tumors? What role does IHC play in NTRK detection, and what are its characteristics? Let’s take a look!

TRK includes three transmembrane neurotrophin receptors: TRKA, TRKB, and TRKC, encoded by the neurotrophic tyrosine receptor kinase genes NTRK1, NTRK2, and NTRK3 located on chromosomes 1q22, 9q21, and 15q25, respectively. In the normal human environment, different TRK protein subtypes bind to different neurotrophic factors, regulating the growth, differentiation, and apoptosis of various neurons in the central and peripheral nervous systems. NTRK fusion refers to: due to intrachromosomal or interchromosomal rearrangements, the NTRK gene encoding TRK fuses with another gene. At this point, the encoded TRK fusion protein is constitutively activated, triggering continuous signal cascades, driving malignant cell proliferation, and leading to cancer.[1]。
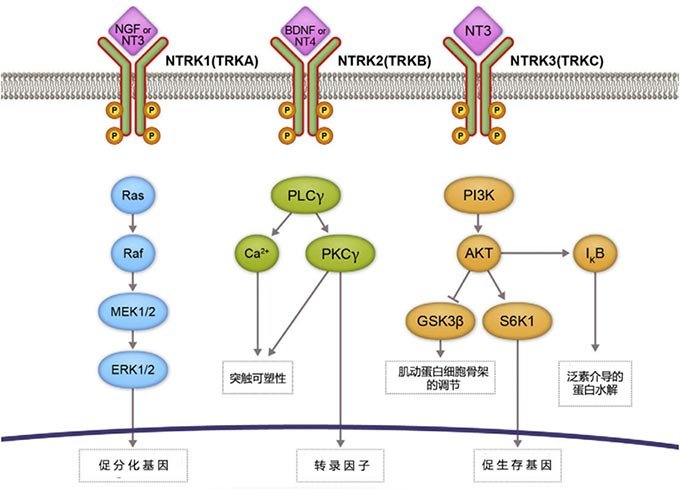
Figure 1. TRK signaling pathway (The binding of neurotrophins to TRK proteins induces receptor dimerization, phosphorylation, and activation of downstream signaling cascades via PI3K, RAS/MAPK/ERK, and PLC-γ)[1]。
NTRK fusion mutations are known as the ‘diamond’ target. Their fusion history can be traced back to 1982 when the NTRK1 gene fusion was first discovered in colon cancer[2]. Subsequently, NTRK fusions have been gradually identified in various adult/child solid tumors.
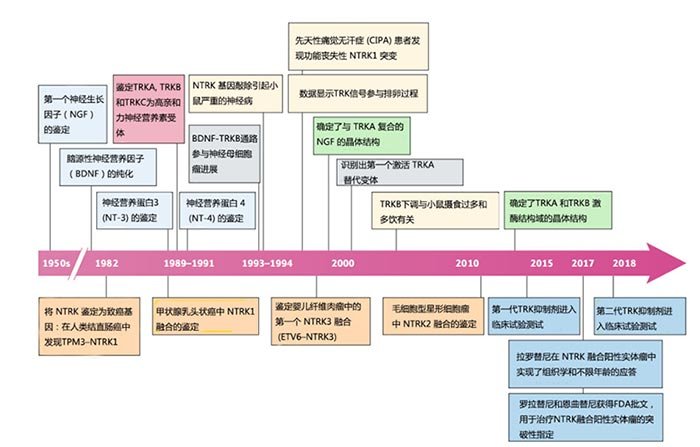
Figure 2. Timeline of key developments related to the biology and therapeutic targeting of TRK signal transduction[3]。
Epidemiological statistics on NTRK fusions reveal[4]that the overall positive rate for NTRK fusion solid tumors is 0.3%. The NTRK fusion positive rates for adult patients (≥18 years) and pediatric patients (<18 years) are 0.28% and 1.34%, respectively; the incidence increases with decreasing age, with the highest NTRK fusion positive rate of 2.28% in children under 5 years old. Among Chinese (or Asian) cancer patients, the NTRK fusion positive rate is 0.4%.
Based on the detected frequency of NTRK fusions, related tumors can be divided into the following two major categories:
I. Rare cancer types highly enriched with NTRK fusions: These include adult secretory carcinoma of the breast, salivary gland carcinoma resembling mammary secretory carcinoma (MASC), and pediatric congenital mesoblastic nephroma (cellular or mixed subtype) and infantile fibrosarcoma, etc., belonging to the ETV6–NTRK3 fusion type, with a prevalence >90% in specific patient series.[3];
II. NTRK fusions occur at lower frequencies in other common tumors (5-25% or <5%): The incidence of NTRK fusion mutations is 5-25% in cancers such as thyroid cancer, Spitzoid tumors, gastrointestinal stromal tumors, and certain pediatric gliomas. The incidence of NTRK fusions is <5% in lung cancer, pancreatic cancer, head and neck squamous cell carcinoma, cholangiocarcinoma, breast cancer, colorectal cancer, renal cell carcinoma, melanoma, adult primary brain tumors (such as astrocytoma or glioblastoma), and non-GIST soft tissue sarcomas. Additionally, some hematological malignancies, such as acute lymphoblastic leukemia and acute myeloid leukemia, also show low-frequency NTRK fusions.[3]。
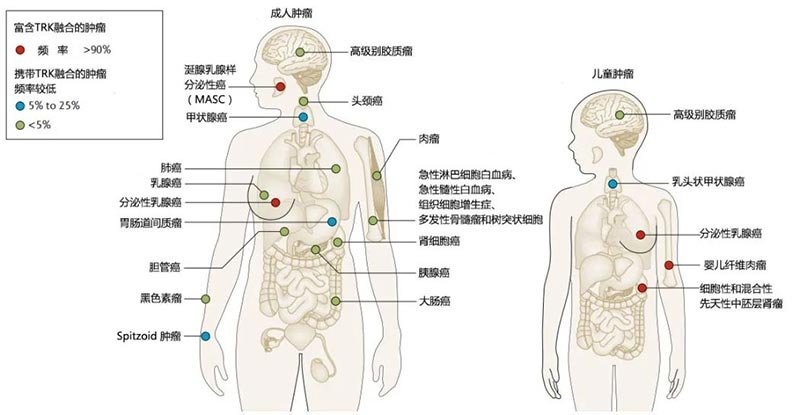
Figure 3.Distribution and frequency of NTRK fusions in adult and pediatric tumors[3]。
Currently, there are more than a dozen clinical-stage investigational new drugs targeting NTRK fusion genes, all possessing TRK inhibitory activity. They can be divided into selective (larotrectinib, selitrectinib, etc.) and non-selective (entrectinib, repotrectinib, etc.) categories.[5]。
Among them, the first-generation oral highly selective TRK inhibitor, larotrectinib, received drug registration approval from the China National Medical Products Administration (NMPA) on April 13, 2022, for the treatment of adult and pediatric patients with solid tumors harboring NTRK fusion genes (including NTRK1, NTRK2, NTRK3). On July 29 of the same year, an oral pan-TRK inhibitor targeting TRKA, TRKB, TRKC, ROS1, and ALK genes, entrectinib, also received NMPA drug registration approval for the treatment of adult and pediatric patients aged 12 and above with NTRK fusion-positive, locally advanced or metastatic solid tumors after initial therapy. This provides more treatment options for tumor patients carrying NTRK fusions. Therefore, detection is crucial for both rare tumors with high-frequency NTRK fusions and common tumors with rare NTRK fusions.
Diagnostic methods for NTRK fusions include IHC, FISH, PCR, and NGS. Among these, IHC offers advantages of high sensitivity, strong specificity, short processing time, and low cost.[6]。
Research by Danielle A. Murphy et al. found that when using NGS as the reference standard for gene rearrangement detection, pan-TRK IHC demonstrated 100% positive agreement and a 100% negative predictive value, making it an effective method for studying gene fusions.[7]。Consistent with this, Jaclyn F. Hechtman et al. from MSKCC validated pan-TRK IHC on 21 NTRK fusion-positive and 20 negative samples detected by NGS, finding a sensitivity of 95.2%, specificity of 100%, positive predictive value as high as 100%, and negative predictive value as high as 96%.[8]。They also summarized three important uses of pan-TRK IHC: (1) Rapid assessment of advanced malignancies that may harbor NTRK fusions to test patient eligibility for targeted therapy with novel pan-TRK inhibitors; (2) As an alternative to NGS for initial evaluation of possible NTRK fusion cases; (3) A method to confirm surgical pathological diagnosis of specific tumors characterized by NTRK fusions, such as secretory carcinoma of the breast, salivary gland secretory carcinoma/MASC, and congenital fibrosarcoma, etc.[8]。
With deepening research, the European Society for Medical Oncology (ESMO) Translational Research and Precision Medicine Working Group released the 2019 ESMO NTRK Testing Guidelines, pointing out: For tumors with low-frequency mutations in high-incidence tumor types, a ‘two-step approach’ can be considered, using pan-TRK IHC detection first for enrichment and screening of positive mutations; for tumors with high-frequency mutations in rare tumor types, even if NGS is routinely performed, pan-TRK IHC detection at the protein level is also recommended for cross-validation and data accumulation.[6]。
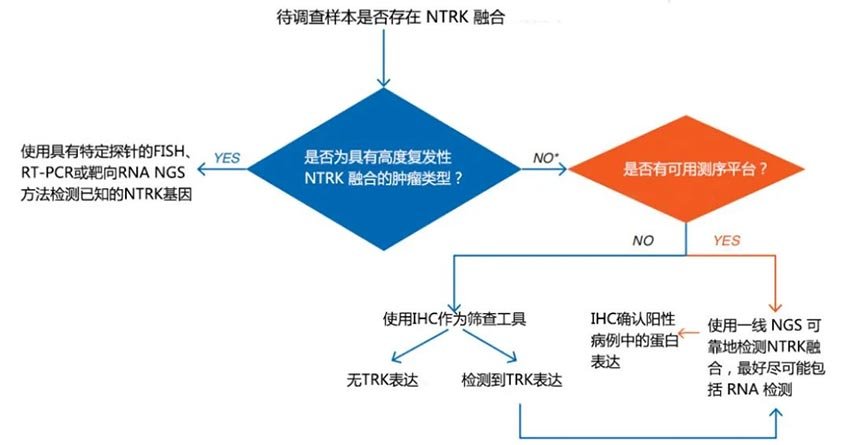
Figure 4. 2019 ESMO recommendations for standard methods to detect NTRK fusions in clinical practice and research[5]。
Furthermore, the recently released ‘Chinese Expert Consensus on Clinical Diagnosis and Treatment of NTRK Fusion Genes in Solid Tumors’ also states: pan-TRK IHC can serve as an initial screening method for cancer types with low incidence of NTRK fusion genes, where NTRK is not expressed and NGS DNA panel methods are not routinely recommended; it can also serve as a verification method for NGS detection.[4]。
Maxin’s self-developed pan-TRK rabbit monoclonal antibody reacts with a conserved proprietary peptide sequence from the C-terminus of TRKA, TRKB, and TRKC, enabling simultaneous recognition of TRKA, TRKB, and TRKC proteins. Therefore, it has the characteristic of covering all fusion types. Throughout the development process, we have always focused on customer needs, established a unique validation standard and quality control system, incorporated IHC validation at every step of screening, and persisted in stability and optimization for compatibility with automated pathology staining systems. It features high sensitivity, high specificity, and high stability, with precise and reliable IHC staining results!
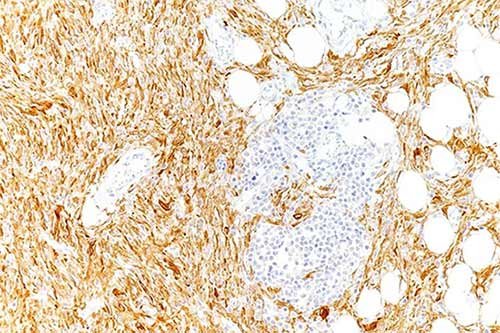
Figure 5. Pan-TRK staining of lipofibromatosis-like neural tumor, cytoplasmic & nuclear positive
|
Antibody Name
|
Product Number
|
Clone Number
|
Cellular Localization
|
|
pan-TRK
|
RMA-1072
|
MXR032
|
Cytoplasm/Nucleus/Membrane
|
[1]Farago, Anna, F, et al.Targeting TRK family proteins in cancer[J]. Pharmacology and Therapeutics: The Journal of the International Encyclopedia of Pharmacology and Therapeutics, 2017, 173:58-66.
[2]Vaishnavi A , Le A T , Doebele R C . TRKing Down an Old Oncogene in a New Era of Targeted Therapy[J]. Cancer Discovery, 2014.
[3]Cocco E , Scaltriti M , Drilon A . NTRK fusion-positive cancers and TRK inhibitor therapy[J]. Nature Reviews Clinical Oncology, 2018, 15:731-747.
[4] Chinese Expert Consensus on Clinical Diagnosis and Treatment of NTRK Fusion Genes in Solid Tumors. http://caclp.com/article/?detail_9916.html.
[5] Qi Changsong, Dong Kun, Yuan Jiajia, et al. Detection of NTRK gene fusions and research progress of TRK inhibitors [J]. Journal of Multidisciplinary Cancer Management (Electronic Edition), 2022, 8(2): 113-121.
[6]C Marchiò, Scaltriti M , Ladanyi M , et al. ESMO recommendations on the standard methods to detect NTRK fusions in daily practice and clinical research[J]. Annals of Oncology, 2019, 30(9).
[7]Murphy D A , Ely H A , Shoemaker R , et al. Detecting Gene Rearrangements in Patient Populations Through a 2-Step Diagnostic Test Comprised of Rapid IHC Enrichment Followed by Sensitive Next-Generation Sequencing[J]. Applied Immunohistochemistry & Molecular Morphology Aimm, 2016.
[8]Hechtman Jaclyn F,Benayed Ryma,Hyman David M,et al. Pan-Trk Immunohistochemistry Is an Efficient and Reliable Screen for the Detection of NTRK Fusions.[J]. The American journal of surgical pathology,2017,41(11).
For more information, please contact: 800-8581156 or 400-889-9853







