Research Project Introduction
This research project involved four major centers from east, west, north, and south: Fudan University Cancer Hospital, Guangdong Provincial People’s Hospital, West China Hospital of Sichuan University, and Cancer Hospital of the Chinese Academy of Medical Sciences.A retrospective study was conducted on218 cases of esophageal squamous cell carcinomawith a time span from June 2020 to June 2021.Simultaneously,Four domestic PD-L1 antibodies including Maixin and their respective platforms were selected for consistency comparison with the imported PD-L1 22C3 pharmDx.

Research Project Background
It is gratifying that among the four antibodies participating in this evaluation, two are Chinese original “clones,” which also marks that domestic enterprises have caught up in the development of companion diagnostic antibodies,Core Technological Innovationlaying the foundation for competing on the same stage with multinational corporations and even overtaking them.
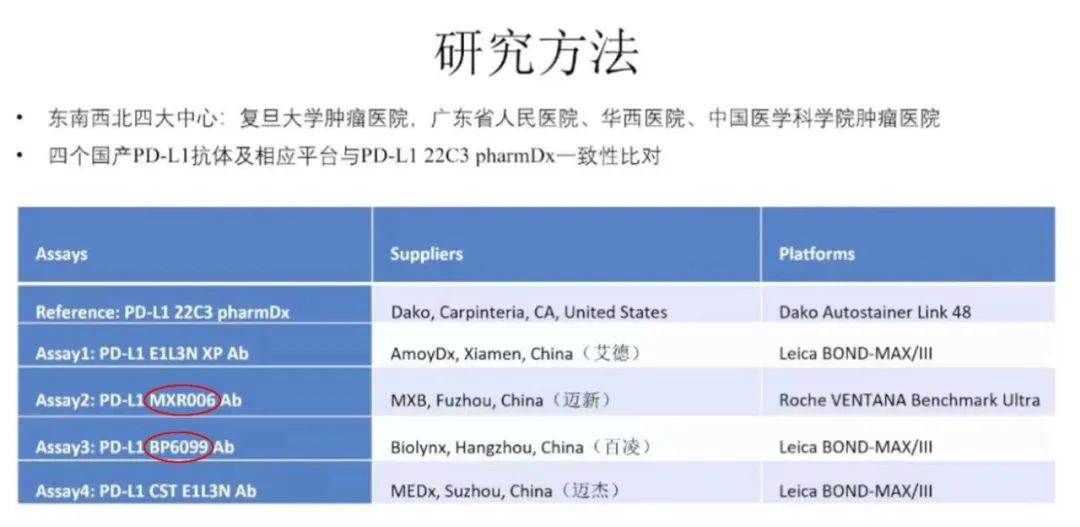
Maixin (MXR006) isPD-L1one of theinChina.two original “clones”
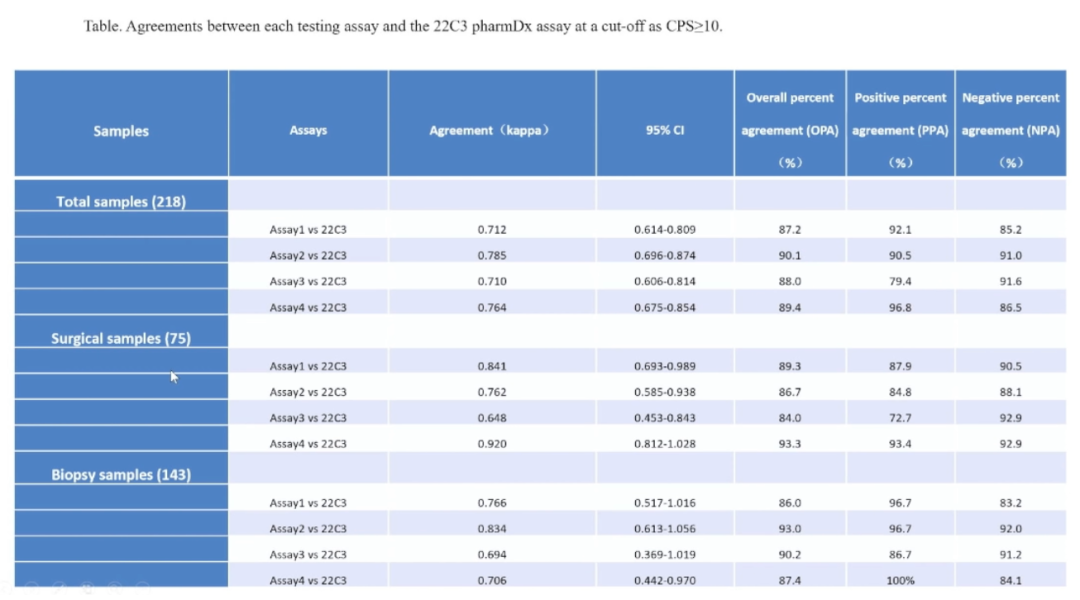
ExperimentUsingTPS (Tumor Proportion Score) and CPS (Combined Positive Score)as continuous variables [bubble plot and Spearman rank correlationcoefficient (p)] for analysis and presentation,each center stained and evaluated the antibodies’TPSand CPS, while using PD-L1 clinically relevant cut-off CPS (≥10) as a binary variable for negative and positive (measured by kappa value to assess consistency), calculating the overall percent agreement (OPA), positive percent agreement (PPA), and negative percent agreement (NPA) between each domestic antibody and22C3 pharmDx.Overall percent agreement(OPA), through a scientifically established evaluation system from different dimensions,comprehensively comparing the consistency of staining between the four domestic antibodies and 22C3 pharmDx.
Statistical Data of Various Evaluation Parameters
Summary of Research Findings
The research results indicate: Maixin PD-L1 antibody performed excellently (with the highest scores in Kappa value, OPA, PPA, etc.), showing good consistency with the imported 22C3 antibody and possessing strong potential application value.
We Have Always Been Striving
Cancer, as a formidable mountain in the history of humanity’s struggle against disease, the traditional trio—surgery, chemotherapy, and radiotherapy—clearlycan no longer meet people’s expectations for tumor treatment.In recent years,Immunotherapy represented by PD-1/PD-L1 immune checkpoint inhibitors has achieved significant success in the clinical application of treating solid tumors such as esophageal cancer and non-small cell lung cancer,bringing new choices and hope for the treatment of tumor patients.but effective immunotherapy requires precise diagnosis. Only through precise pathological detection methods to screen patient groups suitable for immunotherapy can immunotherapy exert its intended efficacy.
Maixin has been deeply engaged innearlyfor thirty years,committed to promoting the development of immunohistochemistry in China.This research achievement fully demonstrates the excellent performance of Maixin PD-L1 antibody, affirming Maixin’s years of commitment to independent innovation, and will profoundly inspire Maixin personnel actively exploring on the scientific path.With the deepening of tumor biology research and the continuous emergence of new mechanisms and targets, we will remember the mission and responsibility of national enterprises, actively advance research in the field of companion diagnostics in the era of immunotherapy, develop more innovative products that keep pace with the times, and demonstrate the “core” strength of domestic pathology enterprises.,Comprehensively assisting in precise tumor immunotherapy,benefiting global tumor patients.





