
If you often walk by the river, how can you avoid getting your shoes wet? Colleagues who have made mistakes know that abnormal expression of cytokeratin can also occur in sarcomas and other mesenchymal tumors. Various misdiagnosis scenarios caused by AE1/AE3 have been proven to be due to the presence of multiple cytokeratin subtypes in non-epithelial tumors. For example, in vascular tumors, CK1 is the main CK subtype expressed; CK1 and CK7 are expressed in some retroperitoneal schwannomas but not in peripheral schwannomas. Synovial sarcoma can express CK7, CK8, CK14, and CK19, with a few cases also expressing CK6, CK13, CK16, CK17, and CK20.
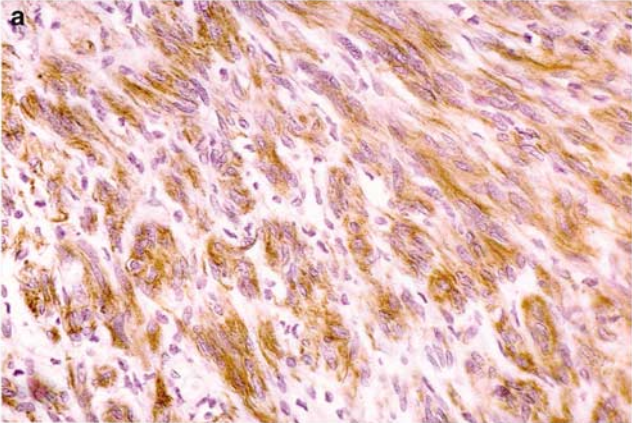
Figure 1. Cited from “Keratin expression in schwannoma; a study of 115 retroperitoneal and 22 peripheral schwannomas”; Schwannoma expresses AE1/AE3.
Epithelioid sarcoma can express CK5/6, CK14, and CK20 to varying degrees depending on the epithelial differentiation of the tumor cells. Meningiomas typically express CK18 in spindle cells adjacent to glandular spaces, while secretory meningiomas consistently express CK19.
Adegboyega performed multiple cytokeratin antibody staining on 17 cases of endometrial stromal sarcoma, with 8 cases showing AE1/AE3 positivity. According to previous reports by some researchers, abnormal cytokeratin expression in sarcomas is weak and focal, limited to a few tumor cells. However, Adegboyega’s study showed that the abnormal expression rate of AE1/AE3 in endometrial stromal sarcoma is close to 50%.
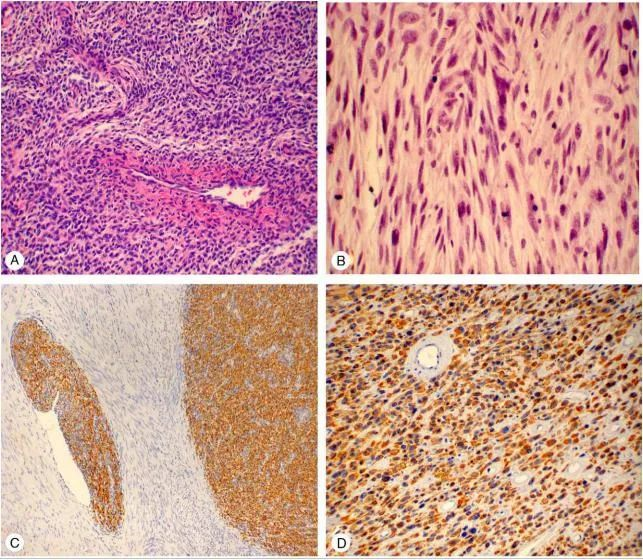
Figure 2. Cited from “Immunohistochemical profiling of cytokeratin expression by endometrial stroma sarcoma”; A and B are endometrial stromal sarcoma, C is low-grade endometrial stromal sarcoma, D is high-grade endometrial stromal sarcoma, and both C and D express AE1/AE3.
Polyostotic fibrous dysplasia, also known as Albright syndrome or McCune-Albright syndrome (MAS). This disease was first described by McCune and Albright and is a bone growth and developmental disorder. Clinically, it is characterized by three main features: bone lesions, precocious puberty, and skin pigmentation. A literature report described a 45-year-old male patient with polyostotic fibrous dysplasia who sought medical attention due to progressive worsening hip pain, limited mobility, and shortening of the left lower limb. Pathological biopsy of the left femur and left tibia showed focal areas of tumor cells expressing AE1/AE3.
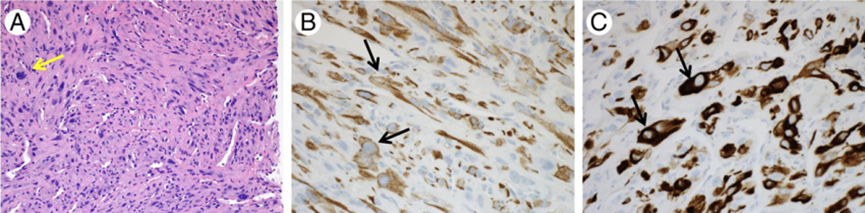
Figure 3. Cited from “Malignant transformation of polyostotic fibrous dysplasia with aberrant keratin expression”; Polyostotic fibrous dysplasia (A) shows a high-grade malignant spindle cell tumor composed of pleomorphic spindle cells; the nuclei have deep chromatin staining, occasional atypical mitotic figures (arrows), and occasionally prominent nucleoli (HE). Focal areas of tumor cells express AE1/AE3 (B) and OSCARC (arrows).
Here, we also introduce AE1/AE3’s sibling—cytokeratin OSCAR, which has a similar expression pattern to CAM5.2 but with a higher molecular weight range (e.g., it can label squamous epithelium). In diagnosis, OSCAR is often used in combination with other antibodies to determine the cell lineage of poorly differentiated malignant tumors.
Since there are focal areas with strong cytokeratin expression in the lesions of fibrous dysplasia, the differential diagnosis of this case needs to exclude metastatic sarcomatoid carcinoma. Based on the patient’s history of polyostotic fibrous dysplasia and comparison of previous and current imaging data, it was found that a new malignant mass appeared within the previously well-defined fibrous dysplasia lesion, and the ground-glass opacity in the proximal left femur had thinned. The final diagnosis of this case was malignant transformation of fibrous dysplasia.
Since some spindle cell lesions can express AE1/AE3, can gastrointestinal stromal tumors (GIST) do so? Sing et al. performed immunohistochemical staining on 64 GIST patients and found that 7 cases expressed AE1/AE3. They found that all 7 patients with AE1/AE3 expression were high-risk GIST, and 4 of them had distant organ metastasis.
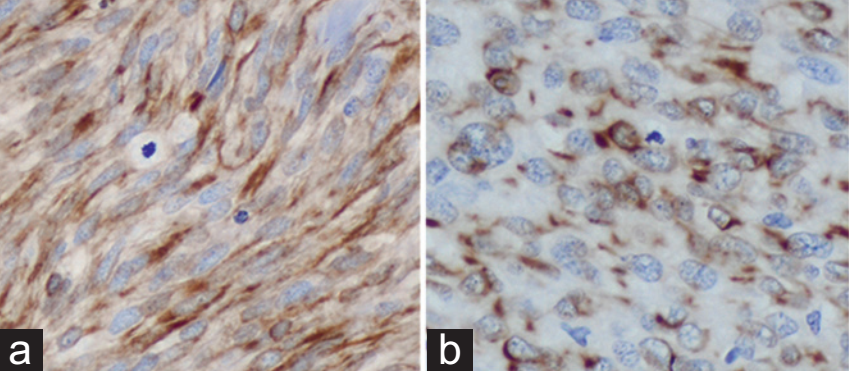
Figure 4. Cited from “Cytokeratin expression in gastrointestinal stromal tumors: morphology, meaning, and mimicry”; The tumor cells are pleomorphic, spindle-shaped and epithelioid, expressing AE1/AE3.
Sarlomo et al. believe that high-risk GIST more frequently expresses AE1/AE3 than low-risk GIST. In their study, the positive rate of AE1/AE3 was 66.7% (8/12). These findings suggest that AE1/AE3 expression in GIST may be a phenomenon related to tumor progression, and AE1/AE3 is common in high-risk GIST; additionally, AE1/AE3 positive expression is also common in metastatic malignant melanoma.
Zhao et al. reported a case of solitary fibrous tumor occurring in the pelvis, with focal positive staining for AE1/AE3. The patient was a 53-year-old female who incidentally discovered a pelvic mass, with the tumor’s maximum diameter being 9 cm.
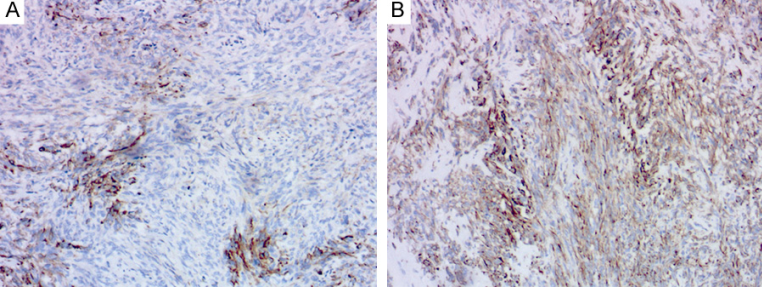
Figure 5. Cited from “A pelvic cellular solitary fibrous tumor with multifocal expression of cytokeratin AE1/AE3”; Solitary fibrous tumor, Figures A and B show focal positivity for AE1/AE3.
In addition to solitary fibrous tumors, abnormal AE1/AE3 expression is not uncommon in fibroblastic/myofibroblastic soft tissue lesions, such as inflammatory myofibroblastic tumor and proliferative fasciitis. In a study by Barak et al., 3 out of 27 cases of solitary fibrous tumors (11%) showed focal AE1/AE3 positivity. According to Stephanie, the positive rate of AE1/AE3 in proliferative fasciitis is 46% (6/13).
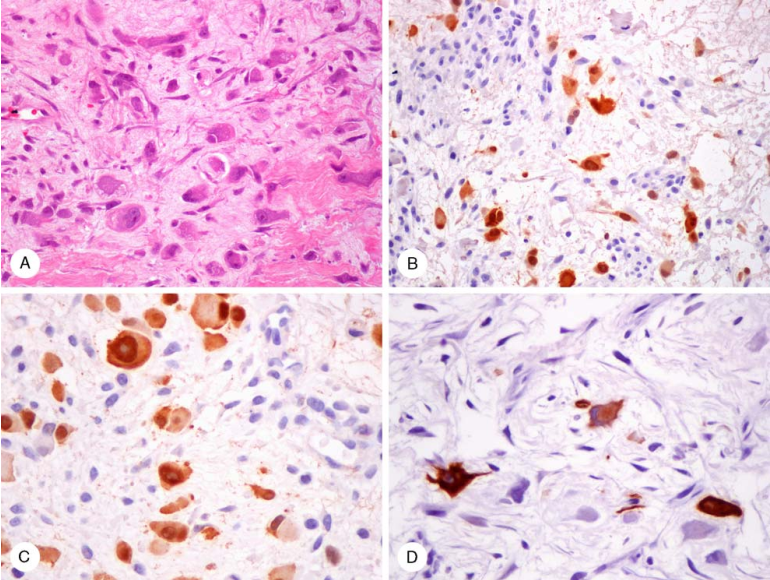
Figure 6. Cited from “Immunoreactivity for calretinin and keratins in desmoid fibromatosis andother myofibroblastic tumors”; A, Ganglion-like cells in proliferative fasciitis. B and C, Cytoplasm and nuclei of ganglion-like cells in proliferative fasciitis show calretinin positivity. D, Ganglion-like cells are AE1/AE3 positive, with immunostaining highlighting their dendritic appearance.
The presence of cytokeratin immunoreactivity in some myofibroblastic tumors may mislead many novices to interpret this finding as evidence of epithelial differentiation, especially when facing calretinin-positive tumors, which should raise suspicion of mesothelioma.
AE1/AE3 may show abnormal staining in gliomas (ependymoma, glioblastoma, schwannoma).To this end, Bacchi et al. conducted a series of experiments and observed that AE1/AE3 staining could only be detected in formalin-fixed, paraffin-embedded normal brain tissue and gliomas after enzyme digestion, but not in tissues fixed with methoxyacetanilide.
AE1/AE3 positive expression can also be seen in some hematopoietic system diseases. Some plasma cell lesions can show focal positivity for multiple epithelial markers, with EMA positivity rate about 50%, high molecular weight CK 14%, and broad-spectrum cytokeratin AE1/AE3 7%. Some plasma cell lesions show Golgi dot-like positivity for high molecular weight CK and AE1/AE3, while EMA is expressed in the cytoplasm. Diffuse AE1/AE3 positivity is rarely reported in the literature. Experienced pathologists can use CD38, CD138, and MUM-1 in combination when diagnosing AE1/AE3-positive plasma cell lesions to avoid misdiagnosis.
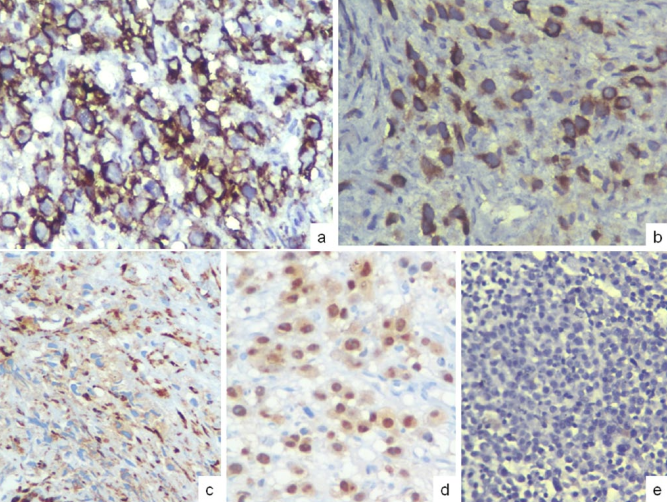
Figure 7. Cited from “Cytokeratin expression in plasmacytomas: a comprehensive analysis using modern cytokeratin cocktails”; Plasmacytoma, a 51-year-old male patient who had received chemotherapy with cyclophosphamide, bortezomib, lenalidomide, and dexamethasone, showed immunohistochemical AE1/AE3 positivity after chemotherapy (a) diffuse CD138 membrane positivity, (b) AE1/AE3 focal strong cytoplasmic staining, (c) CD38 shows granular cytoplasmic staining, (d) MUM-1 shows about 40% nuclear positivity, (e) plasma cells in the patient’s bone marrow before chemotherapy showed AE1/AE3 negativity.
Lymphoma patients can also show AE1/AE3 positive expression. Coulibaly reported a case of a 51-year-old male patient with intravascular diffuse large B-cell lymphoma, with unknown past medical history, who developed pain in the right S1 nerve root 4 weeks prior and left L4 skin paresthesia. The patient subsequently underwent biopsy of an adrenal mass.
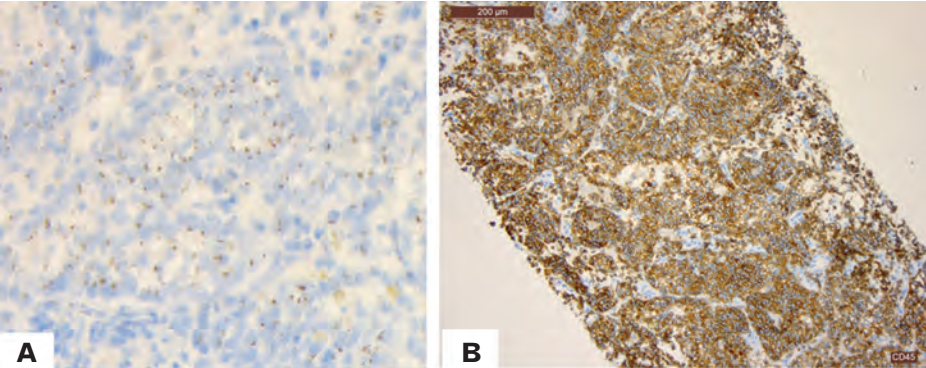
Figure 8. Cited from “Intravascular large B-cell lymphoma presenting as cauda equina syndrome and showing aberrant cytokeratin expression: a diagnostic challenge”; Intravascular diffuse large B-cell lymphoma, (A) tumor cells show abnormal dot-like staining for AE1/AE3. (B) CD45 positive.
This is also the first report in the literature of intravascular diffuse large B-cell lymphoma expressing AE1/AE3, but the reason for its expression and this unique dot-like staining pattern are difficult to explain. Misinterpreting the AE1/AE3 expression in this case could lead to pitfalls. Additionally, this dot-like staining pattern of AE1/AE3 can also be seen in small cell carcinoma and olfactory neuroblastoma.
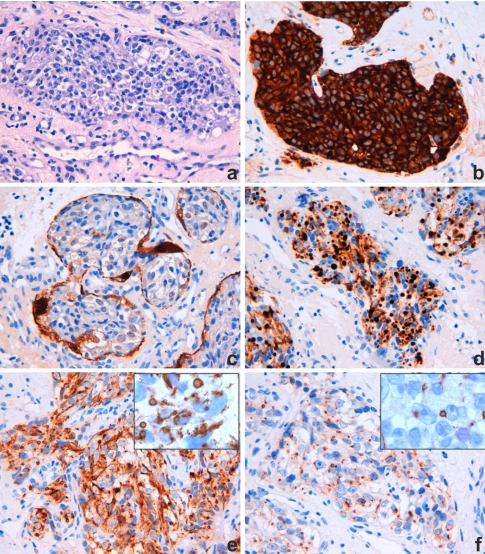
Figure 9. Cited from “Aberrant Pattern of Cytokeratin Expression in Olfactory Neuroblastoma: A Potential Diagnostic Pitfall”; (a) Olfactory neuroblastoma, HE staining; (b) syn; (c) S100; (d) cytokeratin combined labeling (CK5/6/8/18); (e) CK18; (f) CK8.
For example, Mandaran et al. reported a 44-year-old male patient with olfactory neuroblastoma. Right nasal biopsy showed an epithelioid tumor infiltrating the submucosa of the nasal cavity, forming nests with rich vasculature. The tumor cells had abundant cytoplasm and atypical nuclei with prominent nucleoli. Mitotic figures were rare. They found that when using cytokeratin markers on this tumor, the tumor cells showed perinuclear dot-like staining.
Fernand reported a 36-year-old woman who underwent a cesarean section for a full-term second child. During the puerperium, a 2.5 cm diameter subcutaneous nodule was found in the abdomen, located at the site of a cesarean section scar from 3 years prior. Seeing this, many might say, isn’t this obviously endometriosis caused by the cesarean section? However, foreigners believe that nodules formed by decidual tissue without an embryo outside the uterus are called deciduomas. They conducted a series of in-depth studies and found that this deciduoma also expresses AE1/AE3.
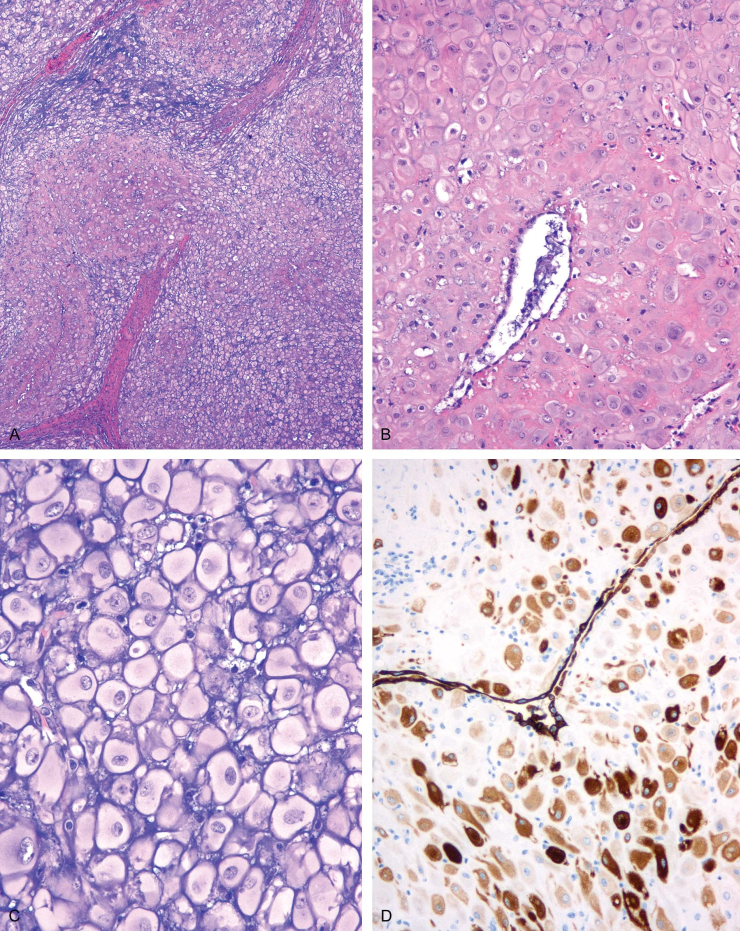
Figure 10. Cited from “Hypodermal decidualized endometrioma with aberrant cytokeratin expression. A lesion mimicking malignancy”; Deciduoma. A, Large cell nests incompletely separated by fibrous septa. There is an inconspicuous glandular cleft at the bottom left. B, Mononuclear epithelioid cells, occasionally with clefts, lined by cuboidal or flattened cells. C, Uniform large polygonal cells with pale, well-defined cytoplasm. The nuclei have fine chromatin and prominent nucleoli. Extracellular basophilic stroma is present. D, Scattered positivity for low molecular weight cytokeratin.
Yeh et al. reported that AE1/AE3 can be used to distinguish decidua from intermediate trophoblast cells. Other literature indicates that decidualized cells in or outside the uterus can express AE1/AE3, CK18, and inhibin-α. This deciduoma case needs to be differentiated from deciduoid mesothelioma. This disease occurs in the peritoneum of young women who have never had asbestos exposure, may occur after cesarean section, and may be associated with hCG secretion. Deciduoid mesothelioma tumor cells are large, round, oval, or polygonal. The tumor cells have abundant ground-glass eosinophilic cytoplasm, clear cytoplasmic boundaries, round nuclei, and prominent eosinophilic nucleoli. Some areas of the lesion may show gland-like and papillary structures. Additionally, immunohistochemistry can express calretinin, AE1/AE3, CK5/6, and human mesothelial cell antigen-1. Most patients with this rare tumor die within a year.
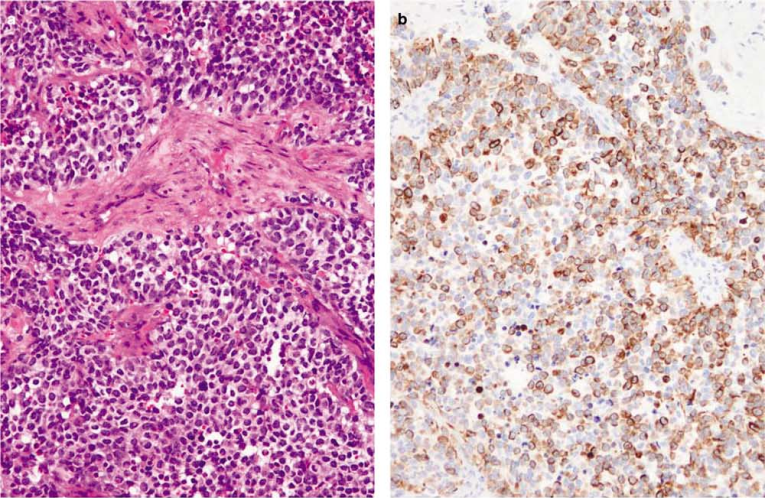
Figure 11. Cited from “Aberrant expression of epithelial and neuroendocrine markers in alveolar rhabdomyosarcoma: a potentially serious diagnostic pitfall”; Alveolar rhabdomyosarcoma (a) abnormal expression of AE1/AE3 (b).
AE1/AE3 expression is also common in alveolar rhabdomyosarcoma, with a positive rate of 30%-40%. This finding has certain diagnostic significance for alveolar rhabdomyosarcoma, especially when it occurs in the head and neck region of adults, requiring careful diagnosis. Desmoplastic small round cell tumor and Wilms tumor can also express AE1/AE3. Granja et al. studied 44 cases of desmoplastic small round cell tumor and found that nearly 50% of cases expressed broad-spectrum cytokeratin or low molecular weight cytokeratin. Additionally, yolk sac tumor can also express AE1/AE3.
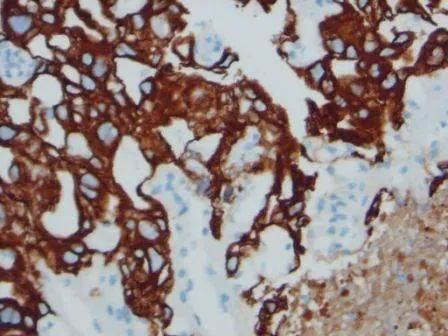
Figure 12. Cited from “Yolk Sac Tumor in the Anterior Mediastinum: A Rare Case Matthew Coleman”; The figure shows yolk sac tumor AE1/AE3 positive.
AE1/AE3 positive expression can also occur in undifferentiated sarcoma, osteosarcoma, adenomatoid tumor, adenomatoid odontogenic tumor, aggressive angiomyxoma, angiosarcoma, chondroblastoma, chordoma, choroid plexus tumor, ependymoma, epithelioid sarcoma, gastrointestinal reactive nodular fibrous pseudotumor, glioblastoma, glioma, epithelioid hemangioendothelioma, hepatoblastoma, etc. Therefore, for the ubiquitous AE1/AE3, we must always be vigilant in diagnostic work to avoid pitfalls.
|
Antibody Name
|
Product Number
|
Clone Number
|
Cellular Localization
|
|
CK(Pan)
|
MAB-0671
|
MX005
|
Cytoplasm
|
|
CK(Pan)
|
Kit-0009
|
AE1/AE3
|
Cytoplasm
|
References:
1、Bharani V, Sharma P, Bal A, Prakash G. A Plasma Cell Myeloma With Post-Therapy Anaplastic Morphology, Osteomyelosclerosis, and Strong Pan-Cytokeratin (AE1/AE3) Expression: A Potential Diagnostic Pitfall. Int J Surg Pathol. 2018 May;26(3):232-235
2、Yeh I-T, O´ Connor DM, Kurman RJ. Intermediate trophoblast: further immunocytochemical characterization. Mod Pathol. 1990;3:282–287
3、Heatley MK, Maxwell P, Toner PG. The immunophenotype of human decidua and extra-uterine decidual reactions. Histopathology. 1996;29:437–442.
4、Litzky LA, Brooks JJ. Cytokeratin immunoreactivity in malignant fibrous histiocytoma and spindle cell tumors: comparison between frozen and paraffin-embedded tissues. Mod Pathol 1992;5:30-4
5、Granja NM , Begnami MD, Bortolan J, Filho AL, Schmitt FC. Desmoplastic small round cell tumour: Cytological and immunocytochemical features. Cytojournal. 2005 Mar 18;2(1):6.
6、Sarlomo-Rikala M, Tsujimura T, Lendahl U, Miettinen M. Patterns of nestin and other intermediate filament expression distinguish between gastrointestinal stromal tumors, leiomyomas and schwannomas. APMIS 2002;110:499-507
7、Fanburg-Smith JC, Majidi M, Miettinen M. Keratin expression in schwannoma; a study of 115 retroperitoneal and 22 peripheral schwannomas. Mod Pathol. 2006 Jan;19(1): 115-21.
For more information, please contact: 800-8581156 or 400-889-9853













