The INI-1 gene is located at 22q11.2, also known as hSNF5, SMARCB1, and BAF47. It is a core subunit of the SWI/SNF complex, involved in ATP-dependent chromatin remodeling, thereby regulating gene expression and associated with various cellular functions, including repairing damaged DNA and regulating cell growth. The INI-1 protein is considered a tumor suppressor that can prevent rapid cell proliferation. Inactivation of the INI-1 gene leading to loss of protein expression is associated with various malignant tumors, including malignant rhabdoid tumors, epithelioid sarcoma, epithelioid malignant peripheral nerve sheath tumor, renal medullary carcinoma, malignant rhabdoid tumors of the kidney and soft tissues, myoepithelial carcinoma, sinonasal carcinoma, thyroid carcinoma, etc.
INI-1-deficient sinonasal carcinoma is a recently reported rare and highly malignant tumor, accounting for 3%~6% of all primary malignant epithelial tumors of the sinonasal tract. The age of onset ranges from 19 to 89 years, presenting as four subtypes: basaloid, rhabdoid/plasmacytoid, oncocytic, and sarcomatoid.
The study by Shao Liwei et al. indicates that the prognosis of this tumor is worse than that of INI-1-intact sinonasal carcinoma. Misdiagnosis of such tumors should be avoided, and IHC detection of INI-1 aids in the accurate diagnosis of this tumor.
The study by Wang Jiuyang et al. shows that IHC detection of INI-1-deficient sinonasal carcinoma consistently shows negative staining for INI-1. Molecular genetic studies by Agaimy A et al. indicate that 78% of INI-1-deficient sinonasal carcinomas have homozygous or heterozygous deletion of the INI-1 gene. For cases without INI-1 gene deletion, the loss of INI-1 protein expression may be due to epigenetic mechanisms, gene point mutations, or small fragment deletions that cannot be detected.
The study by Shipra Agarwal et al. first reported primary INI-1-deficient thyroid carcinoma. The pathological features of the tumor cells (as shown in Figure 1), IHC shows loss of INI-1 protein expression (a). Furthermore, this study performed INI-1 IHC staining on six other types of thyroid tumors, all showing diffuse strong positive expression (as shown in Figure 2), indicating that INI-1 immunostaining is an effective method for the differential diagnosis of INI-1-deficient thyroid carcinoma.
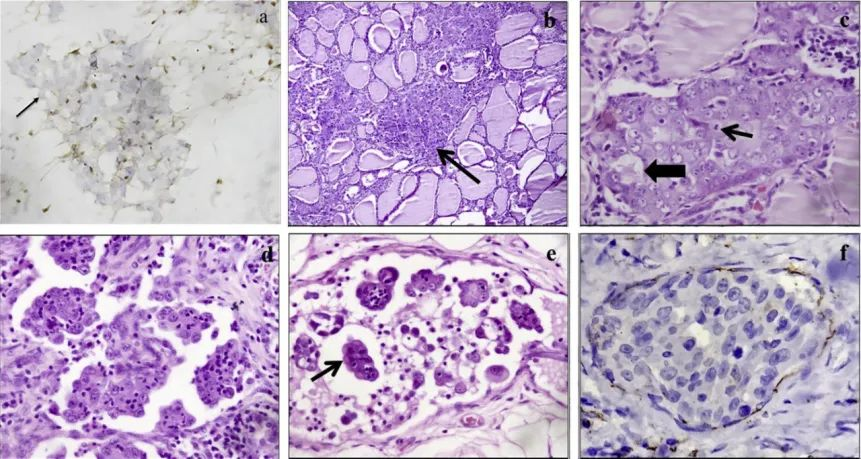
Figure 1. Histopathological features of INI-1-deficient thyroid carcinoma; a: Loss of INI-1 expression by IHC staining; b: Moderate amount of eosinophilic cytoplasm, small and prominent nucleoli. Focal glandular lumen formation is visible; c: Multiple intratthyroidal goiters; d-e: Perithyroidal lymphovascular emboli are visible, with hobnail-shaped cells, lymphocytes, and neutrophils infiltrating within the lesion. A few cells have eccentric nuclei resembling rhabdoid cells. f: CD31 staining confirms perithyroidal lymphovascular emboli.
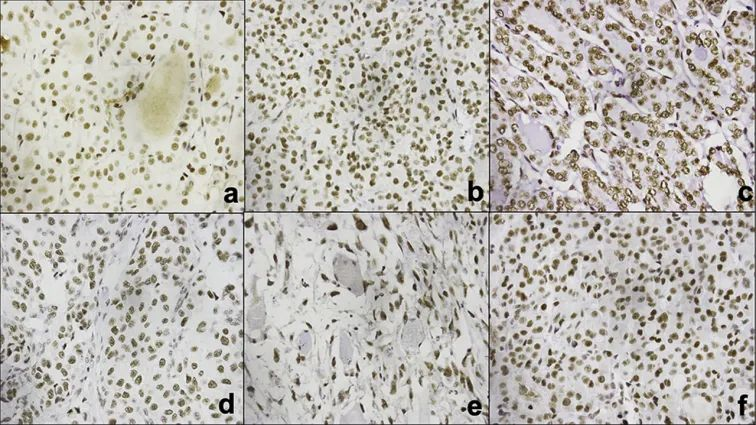
Figure 2. Diffuse positive staining of INI-1 in non-INI-1-deficient thyroid tumors; (A) Follicular adenoma, (B) Follicular carcinoma, (C) Follicular variant of papillary thyroid carcinoma, (D) Poorly differentiated thyroid carcinoma, (E) Anaplastic thyroid carcinoma, and (F) Hurthle cell adenoma.
Malignant Rhabdoid Tumors (MRTs)
Loss or mutation of INI-1 leading to inactivation of both alleles is an important oncogenic mechanism in MRTs. INI-1 IHC can differentiate MRTs from tumors with rhabdoid morphology: Desmoplastic small round cell tumor, rhabdomyosarcoma, and atypical Wilms tumor with rhabdoid morphology all show positive INI-1 IHC staining, while it is negative in MRTs; in synovial sarcoma, INI-1 expression is reduced but not completely lost. Therefore, loss of INI-1 protein expression is an important basis for the diagnosis and differential diagnosis of MRTs.
ES is a malignant mesenchymal tumor with epithelioid cell morphology and immunophenotype, exhibiting diverse morphology. Li Li et al. conducted an IHC study on 20 ES patients and found differences in histomorphology between classical ES and proximal-type ES (as shown in Figure 3, ①-②). The rate of INI-1 loss of expression in the cases was 15/20, with no statistically significant difference between proximal-type and classical ES. INI-1 protein expression pattern was all-or-none, without focal loss (as shown in Figure 3, ③-④). However, many tumors with overlapping morphology with ES, such as vascular tumors, poorly differentiated squamous cell carcinoma, sarcomatoid carcinoma, etc., still retain expression, making it very helpful for the differential diagnosis of ES.
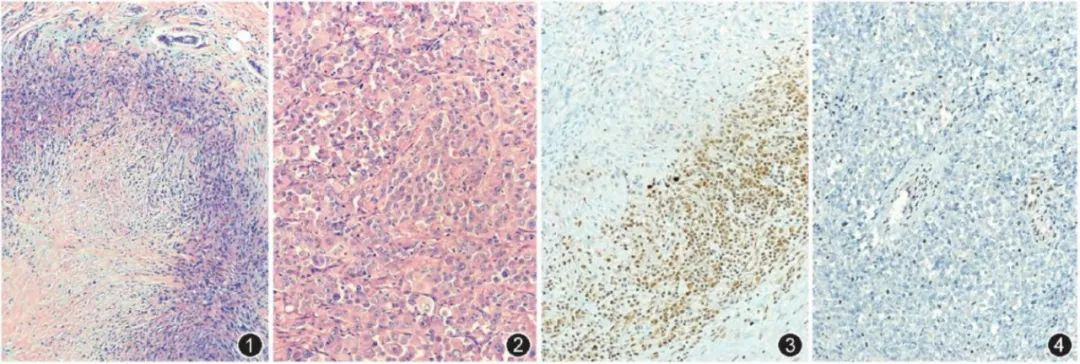
Figure 3. Pathological features of ES ①: Classical ES, tumor cells are spindle-shaped, forming granuloma-like structures with central necrosis and degeneration; ②: Proximal-type ES, tumor cells are diffusely sheet-like, with rhabdoid morphology; ③: Classical ES, INI-1 staining, tumor cell nuclei are negative, surrounding lymphocytes and fibroblast nuclei are positive, EnVision method, medium magnification. Figure 4: Proximal-type ES, INI-1 staining, tumor cell nuclei are negative, surrounding fibroblast and vascular endothelial cell nuclei are positive, EnVision method, medium magnification.
Additionally, the study by Oda Y et al. indicates that the rate of INI-1 loss of expression in ES is 62.5% to 93.0%, making it an indispensable diagnostic indicator.
Epithelioid Malignant Peripheral Nerve Sheath Tumor
Epithelioid malignant peripheral nerve sheath tumor is often composed of uniform epithelioid Schwann cells, often connected to nerves, arising on the basis of neurofibroma or schwannoma. The epithelioid cells within the tumor grow in nests, nodules, or cords, separated by fibrous septa. The tumor cells are uniform in morphology, round or polygonal, with round vesicular nuclei, prominent nucleoli, and rhabdoid cells may be seen. Clinically, differentiation from malignant melanoma is important. The study by Hornick J L et al. shows that 50% of epithelioid malignant peripheral nerve sheath tumor cases show negative INI-1 IHC staining, while malignant melanoma and metastatic carcinoma are positive. Therefore, INI-1 IHC staining is very helpful for the differential diagnosis of difficult cases.
Poorly Differentiated Chordoma
The WHO (2016) classification of central nervous system tumors lists poorly differentiated chordoma as a new tumor type. It occurs predominantly in children, is highly aggressive, and has a poor prognosis, with relatively unique clinicopathological features and immunophenotype. Cells are distributed in lobules or solid sheets, most tumors (79%) lack large vacuolated cells, and there is no extracellular myxoid or chondroid matrix. 96% of cases show loss of nuclear expression of INI-1 by IHC, which is significantly different from other types of chordoma. Therefore, loss of INI-1 expression can serve as a differential diagnostic indicator between poorly differentiated chordoma and classical chordoma.
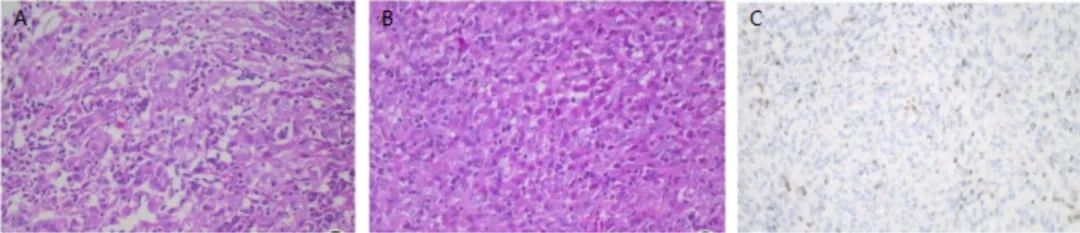
Figure 4. Pathological features of poorly differentiated chordoma A-B: Tumor cells are distributed in sheets, with vesicular nuclei, prominent nucleoli, and significant cellular atypia; C: Loss of INI-1 expression in tumor cells, EnVision method, medium magnification.
The study by Antonelli et al. indicates that loss of INI-1 expression is associated with the poorly differentiated histomorphology and poor prognosis of this tumor, making it one of the prognostic and targeted therapy indicators for INI-1.
Renal Medullary Carcinoma
Renal medullary carcinoma is often associated with sickle cell disease or sickle cell trait. Microscopically, cribriform structures are most common, with reticular, yolk sac-like, solid, sarcomatoid, and adenoid cystic structures also visible. Morphologically, it resembles urothelial carcinoma of the renal pelvis and renal cell carcinoma. However, urothelial carcinoma of the renal pelvis and renal cell carcinoma show positive INI-1 IHC staining, while renal medullary carcinoma shows negative INI-1 IHC staining. Therefore, INI-1 IHC staining can be used for the differential diagnosis of renal medullary carcinoma versus urothelial carcinoma of the renal pelvis and renal cell carcinoma.
Other studies indicate that loss of INI-1 expression also occurs in 40% of pediatric soft tissue myoepithelial carcinomas, 10% of adult soft tissue myoepithelial carcinomas, and a few gastrointestinal tumors and endometrial carcinomas, and may be associated with tumor malignancy and poor prognosis.
Furthermore, novel EZH2 inhibitors as targeted drugs have entered early clinical trial stages for INI-1-deficient tumors. Therefore, IHC detection of INI-1 is a key indicator for targeted therapy.
In summary, loss of INI-1 expression can occur in malignant rhabdoid tumors, epithelioid sarcoma, epithelioid malignant peripheral nerve sheath tumor, renal medullary carcinoma, malignant rhabdoid tumors of the kidney and soft tissues, myoepithelial carcinoma, extraskeletal myxoid chondrosarcoma, sinonasal carcinoma, thyroid carcinoma, and a few gastrointestinal tumors, endometrial carcinomas, etc. It aids in the differential diagnosis of these tumors and is also one of the key indicators for targeted therapy of such tumors.
Related Antibodies from Maixin
|
Antibody Name
|
Product Number
|
Clone Number
|
Cellular Localization
|
|
INI-1
|
MAB-0696
|
MRQ-027
|
Nuclear
|
References
[1] Hornick J L , Cin P D , Fletcher C . Loss of INI1 Expression is Characteristic of Both Conventional and Proximal-type Epithelioid Sarcoma[J]. American Journal of Surgical Pathology.
[2] Antonelli M , Raso A , Mascelli S , et al. SMARCB1/INI1 Involvement in Pediatric Chordoma: A Mutational and Immunohistochemical Analysis[J]. The American journal of surgical pathology, 2017, 41(1):56-61.
[3] Yeter H G , Kosemehmetoglu K , Soylemezoglu F . Poorly Differentiated Chordoma: Review of 53 Cases[J]. Apmis, 2019, 127(9).
[4] Agarwal S , Kakkar A , Damle N A , et al. SMARCB1 (INI1)-deficient thyroid carcinoma: A novel entity expanding the spectrum of tumors with INI1 loss[J]. Pathology – Research and Practice, 2020, 216(4):152830.
[5] Li Li, Zhou Xiaojun. Clinicopathological features of INI1-deficient tumors[J]. Chinese Journal of Pathology, 2015(5):361-364.
[6] Wang Jiuyang, Bai Yuping, Xing Li, et al. Clinicopathological features of SMARCB1(INI1)-deficient sinonasal carcinoma[J]. Chinese Journal of Pathology, 2021, 50(11):1240-1245.
[7] Li Li, Xia Qiuyuan, Rao Qiu, et al. Immunophenotype and INI1 gene alterations in epithelioid sarcoma.[J]. Chinese Journal of Pathology, 2014(6):389-393.
[8] Tao Xuan, Chen Linying, Chen Hong, et al. Clinicopathological and immunohistochemical analysis of epithelioid sarcoma[J]. Journal of Fujian Medical University, 2016, 50(2):98-102
[9] Feng Jiayan, Chen Lian, Ma Yangyang, et al. Clinicopathological observation of poorly differentiated chordoma with INI1 loss[J]. Chinese Journal of Pathology, 2017, 46(10)
[10] Li Sibing, Zhang Yali, Zhang He, et al. Clinicopathological and prognostic analysis of 17 cases of dedifferentiated/undifferentiated endometrial carcinoma[J]. Journal of Clinical and Experimental Pathology, 2021, 37(2):208-213.
For more information, please contact: 800-8581156 or 400-889-9853





