In the previous issue, we outlined the basic categories of renal tumors and the roles of markers such as PAX8, PAX2, CD10, and RCC in the expression of renal tumors. Next, we will explain the different subtypes and immunohistochemistry of five major categories of renal tumors: renal tumors with clear cell morphology, renal tumors with eosinophilic/oncocytic features, tumors with papillary formation, renal medullary carcinoma, collecting duct carcinoma, and other aggressive/invasive tumors. So, in this issue, let’s learn about the large category of renal tumors with clear cell morphology!

Renal Tumors with Clear Cell Morphology
Clear Cell Renal Cell Carcinoma (CCRCC)
CCRCC accounts for nearly 70% of all adult RCCs and is the most clinically relevant RCC subtype. Current treatment guidelines primarily follow a histological binary classification: clear cell and non-clear cell RCC. Although the spectrum of renal tumors with clear cell cytology includes various renal tumors (Table 1), a cautious diagnostic approach is to establish a diagnosis centered on CCRCC.
Table 1. Renal Cell Carcinoma with Clear Cells
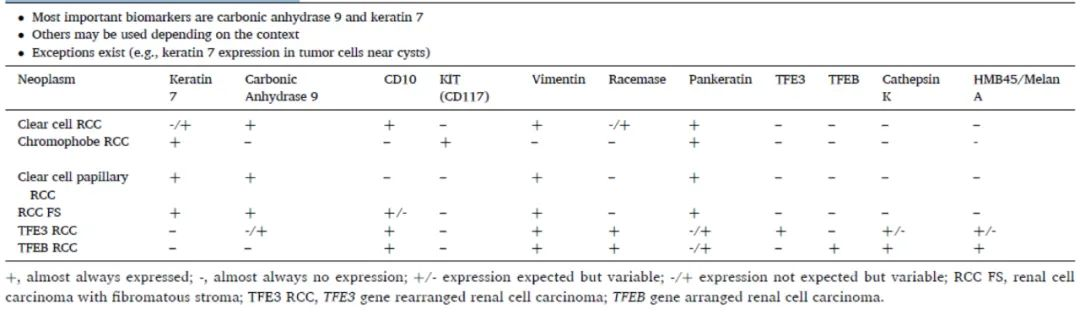
(Click to view larger image)
The histomorphology of CCRCC is shown in Figure 1 (A-I). True papillary formation is extremely rare, but there are subtypes resembling clear cell (tubular) papillary renal cell carcinoma, with branching glandular structures or nuclear arrangements. TFE3-RCC, clear cell papillary RCC, and papillary RCC with clear cell features can be considered tumors with both papillary and clear cell cytological features.
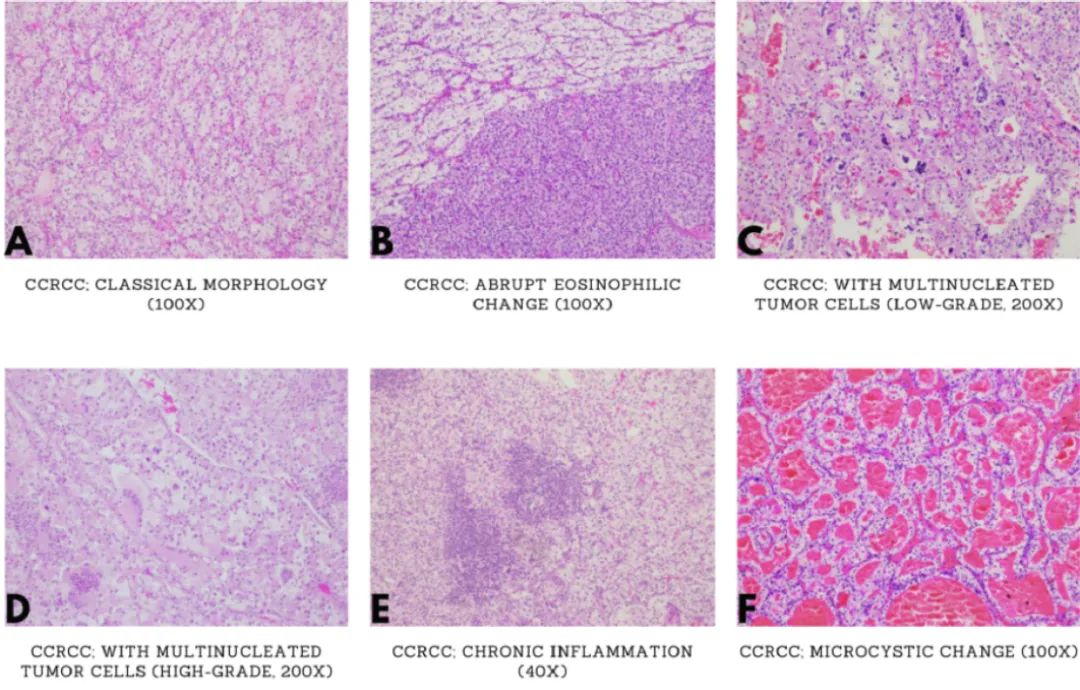
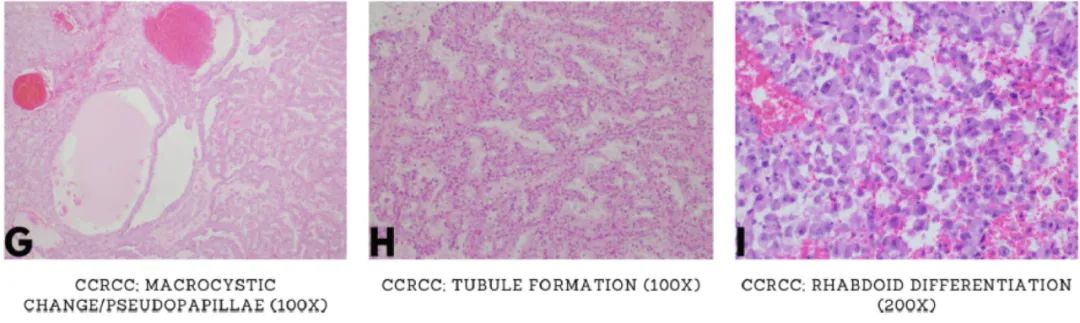
Figure 1. Clear cell renal cell carcinoma (CCRCC) with typical and variant morphologies. (A: Typical CCRCC with abundant cytoplasm, distinct cell borders, arranged in nests or acini, enveloped by a delicate capillary network, but there may be significant heterogeneity within and between tumors; B: Eosinophilic cytoplasm in CCRCC; C and D: Multinucleated giant cells in CCRCC; E: Cytoplasmic or intercellular hyaline deposits, chronic inflammation; F and G: Necrosis, fibrosis/hyalinization, cystic changes, pseudopapillae; H: Tubular structures; I: High-grade features visible)
CA IX shows continuous diffuse and strong membranous staining in over 90% of CCRCCs (Figure 2B), but its expression decreases in high-grade CCRCC. However, CA IX is not specific to RCC; its IHC expression is mainly focal, weak to moderate, and seen in (but not limited to) adrenal glands, esophagus, stomach, colon, prostate, testicular germ cell tumors (especially teratomas), and Reed-Sternberg cells in Hodgkin lymphoma. Besides CCRCC, papillary RCC, FH-RCC, and rare chromophobe RCC also express CA IX, but the distribution is usually not diffuse. Since CA IX is a marker closely related to hypoxia, non-specific expression can be found in viable cells at the edge of necrosis.
CCRCC relatively consistently lacks CK7 expression, except for focal labeling in cystic areas (Figure 2C). Diffuse CK7 labeling is rare in CCRCC. Additionally, CD10 expression is almost always seen in primary and metastatic CCRCC, even in sarcomatoid differentiation. While IHC is unnecessary for diagnosing CCRCC with typical morphology, in ambiguous cases, diffuse and strong membranous CA IX labeling, negative or minimal CK7, and positive CD10 are usually sufficient for diagnosis. KIT, AMACR (Figure 2E), vimentin, and other markers can also be used in specific situations and will be explained in special differential diagnoses.
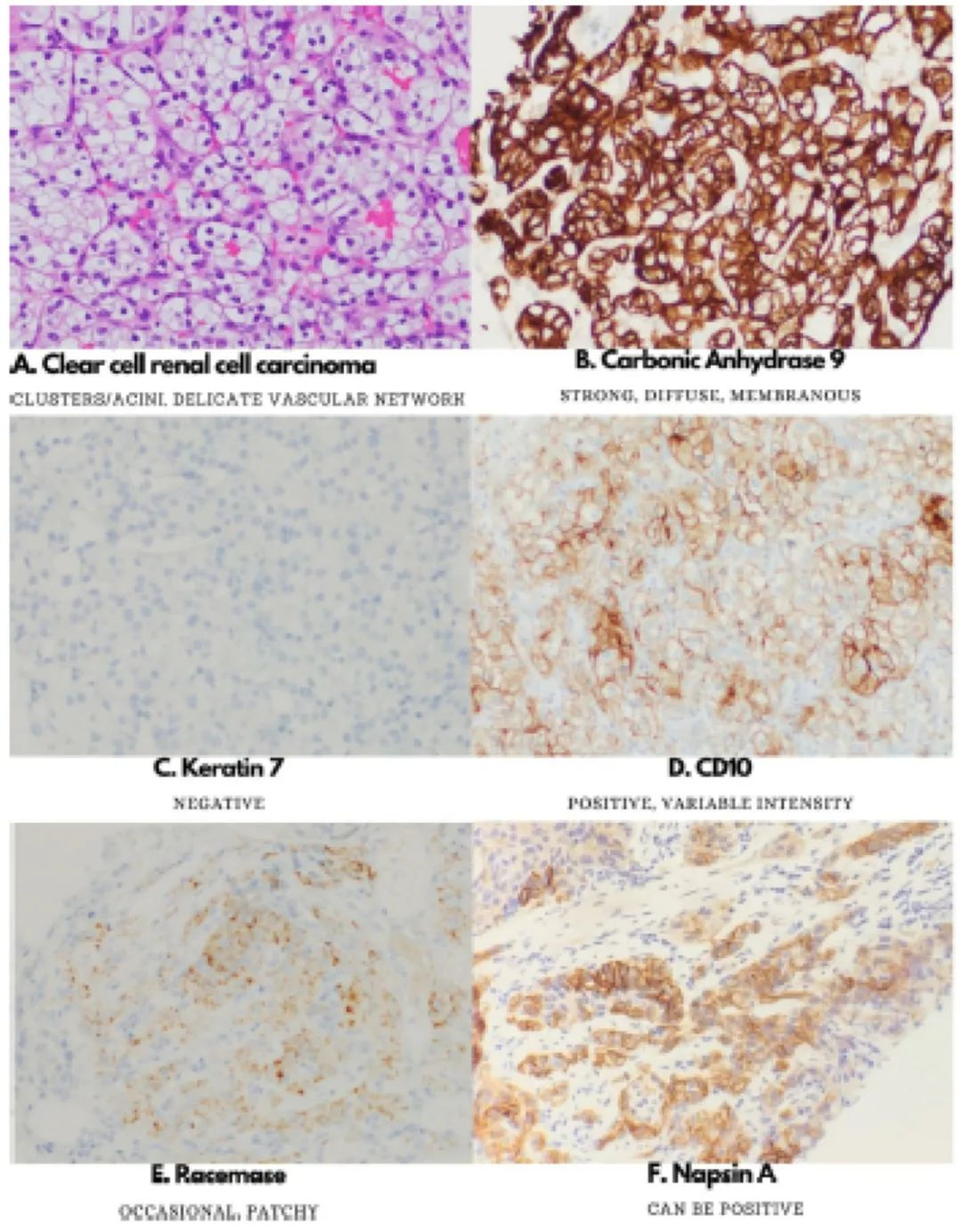
Figure 2. Immunohistochemistry of clear cell renal cell carcinoma.
CCRCC occasionally exhibits aberrant immunohistochemical patterns that may lead to misdiagnosis. About half of CCRCCs express Napsin A, complicating the diagnostic workup for suspected renal or pulmonary metastatic carcinoma (Figure 2F). CCRCC can show TFE3 positivity even in the absence of gene rearrangement, but rarely in a diffuse strong pattern, especially in tumors with high-grade features. A recent analysis of BAP1 indicates that this protein is a recognized poor prognostic marker at both genetic and protein levels. Cases with BAP1 gene mutations and loss of BAP1 protein are associated with distinct histological features, including large eosinophilic cells, high-grade nuclei, and diffuse strong AMACR labeling.
Clear Cell Papillary Renal Cell Carcinoma (CCPRCC)
CCPRCC is a non-aggressive tumor, but its morphology can overlap with CCRCC (Figure 3A), leading to diagnostic difficulties. IHC helps differentiate the two. Compared to CCRCC with diffuse strong CA IX expression, CCPRCC sometimes shows loss of luminal labeling, presenting a so-called cup-shaped pattern (Figure 3B). More importantly, diffuse CK7 expression (Figure 3C) differs from the vast majority of CCRCCs. CD10 (Figure 3D) and AMACR are typically negative in CCPRCC, contrasting with CCRCC and papillary RCC, respectively. Tumors also frequently label for CK-HMW (34βE12) and GATA3, indicating a phenotype more common in the distal nephron than the proximal nephron.
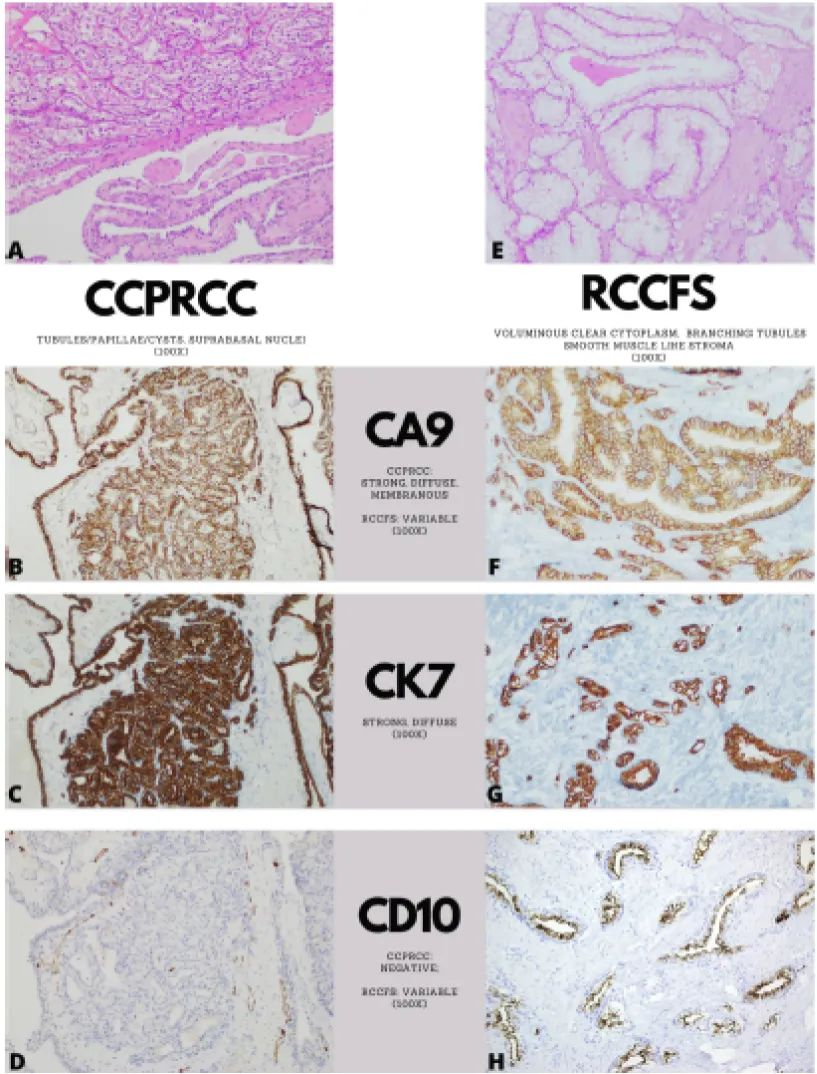
Figure 3. Comparison of morphology and immunophenotype between clear cell papillary renal cell carcinoma (CCPRCC) and renal cell carcinoma with fibromuscular stroma (RCCFS).
Renal Cell Carcinoma with Fibromuscular Stroma (RCCFS)
Recently, renal tumors with larger and clearer cytoplasm forming branching tubules, separated by prominent smooth muscle-like stroma, have been identified with distinct immunohistochemical and molecular features (Figure 3E). RCCFS is considered an inclusive term for these tumors (previously known as leiomyomatous or smooth muscle stroma, among other names). TSC1, TSC2, and MTOR mutations have been found in this tumor, which exhibits variable CA IX (Figure 3F), diffuse CK7 (Figure 3G), and frequent CD10 (Figure 3H) labeling, with minimal AMACR reactivity. RCCFS is often considered to have an indolent outcome, but rare lymph node involvement has been reported in patients with tuberous sclerosis complex. Some clear cell-like tumors with ELOC (formerly TCEB1) mutations have also been recently identified. Compared to CCRCC, these cells show increased CK7 labeling, although the clinical significance of this entity remains controversial and may be difficult to prospectively identify.
MITF Family Translocation RCC
Chromosomal translocations are one of the most common genetic alterations in cancer but are uncommon in renal cell carcinoma. The most common well-defined translocation events in the kidney involve the microphthalmia transcription factor (MITF) family genes TFE3, TFEB, and rarely MITF itself. The most studied is TFE3 gene-rearranged RCC (TFE3-RCC), also the most common translocation-associated RCC, accounting for 40% of pediatric RCCs and 4.2% of adult RCCs. Epithelioid clear to eosinophilic tumor cells form true papillae (Figure 4A), various growth patterns, and occasional psammoma calcifications are considered suggestive morphological features. Occasionally, low-grade oncocytic features (in up to one-quarter of cases) (Figure 4B) or high-grade carcinoma features appear in rare cases.
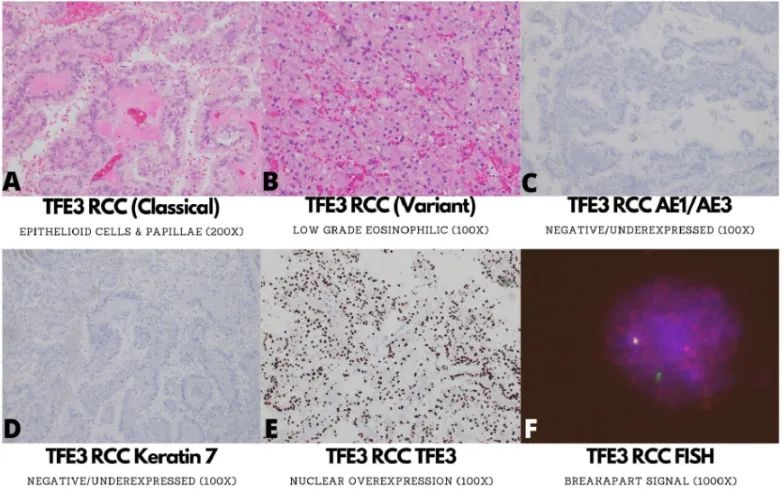
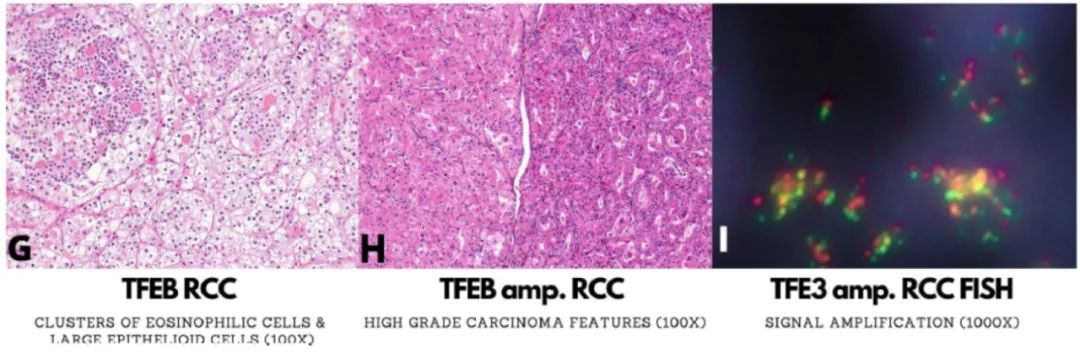
Figure 4. Morphological and immunophenotypic features of TFE3 gene-rearranged renal cell carcinoma (TFE3-RCC), TFEB gene-rearranged renal cell carcinoma (TFEB-RCC), and TFEB-amplified renal cell carcinoma (TFEB amp. RCC).
Initial IHC evaluation and several other relatively large series show minimal or negative keratin labeling (Figure 4C), typically negative CK7 (Figure 4D), positive CA IX, positive AMACR, but negative keratin IHC is not absolute. Melanocytic markers and cathepsin K are variably expressed in TFE3-RCC, especially in patients with PRCC fusions. The most specific IHC marker with the highest positive predictive value for detecting TFE3 FISH rearrangement is TFE3 (Figure 4E). TFE3 IHC expression is also seen in perivascular epithelioid tumors (PEComa) and TMP3-ALK rearranged RCC, with the former having TFE3 gene rearrangement. The current standard for diagnosing TFE3 RCC is primarily FISH (Figure 4F), but some false negatives can occur due to cryptic intrachromosomal translocations.
Among MITF family translocation-associated RCCs, the less commonly rearranged gene is TFEB (TFEB-RCC). The typical morphology initially described for this tumor includes a biphasic cell arrangement with large epithelioid cells similar to TFE3-RCC, surrounded by much smaller clusters of eosinophilic cells with a relatively high nuclear-to-cytoplasmic ratio (Figure 4G). The smaller eosinophilic cells also encircle small spheres of basement membrane-like material. This classic morphological feature is associated with MALAT1-TFEB translocation. Melanocytic markers including HMB45 and Melan-A are frequently positive in TFEB-RCC, and cathepsin-K is positive in almost all TFEB-RCCs. TFEB is overexpressed in TFEB-RCC, although it is also expressed in TFEB-amplified RCC, which is morphologically (Figure 4H and 4I) and clinically distinct from RCC, and renal tumors with high-grade features will be mentioned in this context.
Chromophobe Renal Cell Carcinoma (Chromophobe RCC)
Chromophobe RCC is a relatively common RCC predominantly composed of clear cells, considered to recapitulate intercalated cells of the distal tubule. Its classic type contains two cell populations: large clear cells with prominent cell membranes, resembling plant cell walls; and smaller eosinophilic cells with perinuclear halos (Figure 5A).
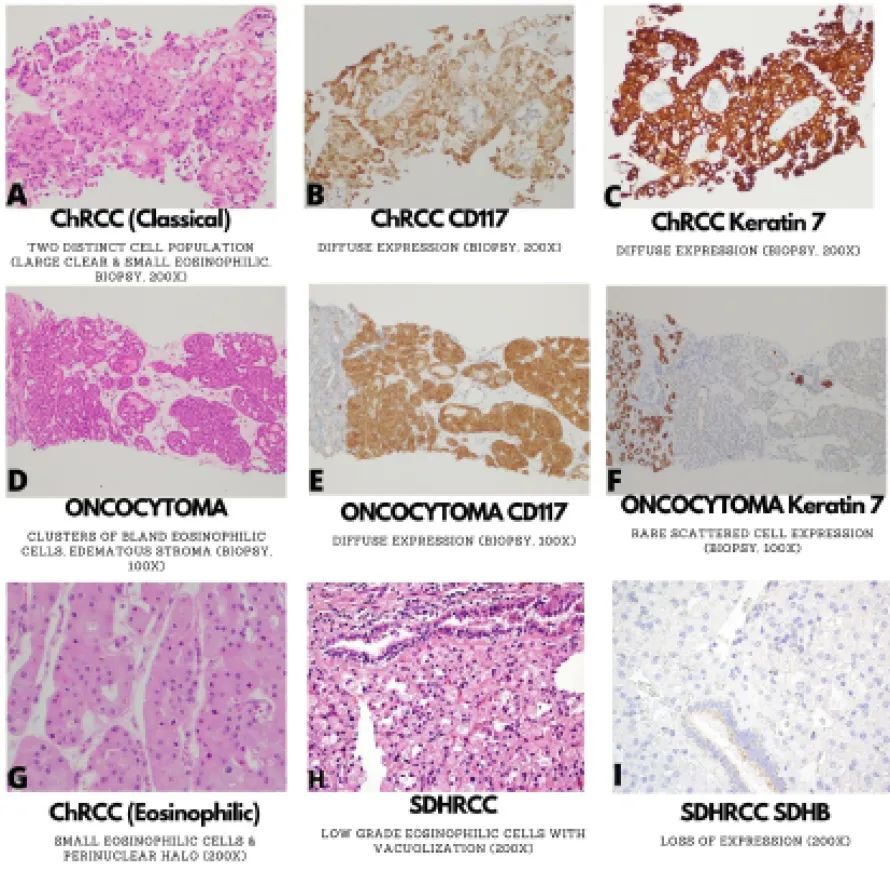
Figure 5. Morphological and immunophenotypic features of chromophobe renal cell carcinoma (typical A-C; eosinophilic variant (G), oncocytoma (D-F), and succinate dehydrogenase-deficient renal cell carcinoma (SDHRCC, H-I).
Both types mentioned above contain nuclei with irregular nuclear membranes.Most chromophobe RCCs show a solid growth pattern with hyalinized vessels, primarily lined by large clear cells (Figure 5A).Diffuse CK7 (Figure 5B) and KIT (Figure 5C) labeling are often seen in chromophobe renal cell carcinoma, which is very rare in other types of clear cell carcinoma.In contrast to CCRCC, vimentin is consistently negative in chromophobe RCC.
Reference: Akgul M, Williamson SR. Immunohistochemistry for the diagnosis of renal epithelial neoplasms. Semin Diagn Pathol, 2022 Jan;39(1):1-16.









